▶️Watch on 3Speak - Odysee - BitChute - Rumble - YouTube - PDF notes
In #FreeEnergy Part 2 I go over a nearly 8-hour crash course in Nuclear Physics, at least the mainstream version of Nuclear Physics, to get an overview of the many definitions and concepts within it. This will help to connect some dots as I research further into the Hidden Science required to develop a free energy device. Once again, the events of 9/11 and the monumental work by Dr. Judy Wood in showing irrefutable proof that the 7 towers turned to dust using advanced top secret free energy tech serves as the motivation and confirmation for me to continue in this quest.
In this overview, I go over the current mainstream view on topics such as:
- Atoms and the Atomic Nucleus
- Nucleons and Isotopes
- Matter and Antimatter
- Quantitation and Quantum Mechanics
- Fundamental Forces in Nature: Gravity, Electromagnetism, Strong Nuclear, and Weak Nuclear
- Subatomic Particles and Elementary Particles
- Magnetic, Electric, and Electromagnetic Fields
- Electromagnetic Radiation, Photons, and Light
- Gamma Radiation and Radioactive Decay
- Nuclear Reactions, Fusion, Fission, and Transmutation
- Nuclear Weapons and the Manhattan Project
- Nuclear Reactors and Fukushima
These are just some of the many topics in nuclear physics that I cover, so if you want to save a year of tuition, make sure to watch this entire video!
Also in this video, I go over the Cold Fusion claims by electrochemists Stanley Pons and Martin Fleischmann, and the subsequent mainstream media, science, and political establishment onslaught and ridicule to coverup what was promising to be a clean free nuclear energy source!
The fall out from this included MIT’s Eugene Mallove resigning in disgust at his school’s role in the Cold Fusion cover up. Mallove later authored both the must-see documentary Cold Fusion: Fire From Water, and the book Cold Fusion: Fire From Ice: Searching for the Truth Behind the Cold Fusion Furor. Later in 2003 he founded the Infinite Energy magazine and website, as well as the New Energy Foundation; both in hopes of waking people up to the possible of free clean energy and towards the developed of this hidden tech. Unexpectedly he was murdered just a year later…
Furthermore, I illustrate the parallels of Dr. Judy Wood’s student Michael Zebuhr and his work in showing that aluminum glows at high heat and normal atmospheric conditions. This was to disprove the almost certain government disinfo agent and founder of the pathetic controlled opposition group Architects & Engineers for 9/11 Truth, Steven Jones, and his claim that aluminum is always silvery in his argument that molten steel flowed from the towers at 9/11. And unexpectedly as well, Zebuhr, whose uncle William (Bill) Zebuhr was the Chairman of Mallove’s New Energy Foundation, was also murdered soon after in 2006. Steven Jones, as I show in the video, was also instrumental in the Cold Fusion coverup; thus making the making 9/11 and Cold Fusion almost inseparable in both the technology used and the means of coverup employed.
On top of that, soon after Michael’s murder Dr. Judy Wood received an email "we've done it before and we will do it again if need be."
And before his death Michael had told Judy that "Whatever happens, don't ever stop pursuing this. It's too important."
It is clear we are up against some pretty evil forces, which is even more reason to keep pursuing game-changing enslavement-ending poverty-ending and conscience-opening free energy technology!
Stay Tuned For #FreeEnergy Part 3…
Free Energy Playlist
View Video Notes Below!
Become a MES Super Fan - Donate - Subscribe via email - MES merchandise
Reuse of my videos:
- Feel free to make use of / re-upload / monetize my videos as long as you provide a link to the original video.
Fight back against censorship:
- Bookmark sites/channels/accounts and check periodically.
- Remember to always archive website pages in case they get deleted/changed.
Recommended Books: "Where Did the Towers Go?" by Dr. Judy Wood
Join my forums: Hive community - Reddit - Discord
Follow along my epic video series: MES Science - MES Experiments - Anti-Gravity (MES Duality) - Free Energy - PG
NOTE 1: If you don't have time to watch this whole video:
- Skip to the end for Summary and Conclusions (if available)
- Play this video at a faster speed.
-- TOP SECRET LIFE HACK: Your brain gets used to faster speed!
-- MES tutorial- Download and read video notes.
- Read notes on the Hive blockchain $HIVE
- Watch the video in parts.
-- Timestamps of all parts are in the description.Browser extension recommendations: Increase video speed - Increase video audio - Text to speech (Android app) – Archive webpages
#FreeEnergy Part 2
Topics to Cover
- Updates
- Revised AI-NWO Idea
- Mainstream Nuclear Physics Overview
- Matter
- Quantum Mechanics
- Fundamental Forces
- Subatomic Particles
- Antimatter
- Electromagnetism
- Radiation
- Nuclear Fission and Fusion
- Nuclear Weapons
- Manhattan Project
- Hiroshima and Nagasaki
- Nuclear Reactors
- Fukushima Disaster
- Cold Fusion
- Pons and Fleischmann
- Eugene Mallove's Murder
- Michael Zebuhr's Murder (Dr. Judy Wood's student)
- Dr. Judy Wood Received Threatening Email
- Summary and Conclusions
Updates
YouTube Unsubscribing My Subscribers??!! # PureEvil
Some of my subscribers told me they weren't getting notifications for my videos for several weeks, only to find out YouTube had unsubscribed them!
I have seen this happen to many other YouTubers and it appears to be just another part of Google/YouTube/CIA suppression of information that is dangerous to the current evil establishment.
In fact, I have noticed my subscriber count gradually increase, but randomly a large junk unsubscribes, and this pattern would repeat itself…
Thus always make sure to BOOKMARK WEBSITES AND PAGES and check periodically yourself!!
Revised AI-NWO Idea
Recall from Part 1 that I speculated as to whether an Advanced Artificial Intelligence (AI) has already taken over utilizing CIA-Blackmail techniques to control politicians.
I realized though that this hypothesis has some difficult to comprehend assumptions such as the powers-that-be, whom have already shown to possess advanced hidden technology and global human perception management tools, would be foolish enough to "lose control" of AI tech…
Thus the push for AI and Blockchain technology at all levels of society may in fact be just part of the overall theme of streamlining the centralization of control.
Thus AI and Blockchain tech should be viewed simply as technology that lowers the number of people required for any given task; such as lowering middlemen, banks, and bureaucracy.
While these technologies have the potential to centralize power, if used properly and democratically can be used to establish fair and transparent forms of governance.
Thus the overall fear of AI taking over the world and a NWO cryptocurrency might just be part of a Psychological Operation to make people feel this is inevitable and to fear these technologies.
But as I argued in # 911Truth Part 5 (https://mes.fm/911truth-playlist), the "NWO" is already here, and their main tool of control is to trickle down suppressed technology to better "steer" the global population.
Technologies such as Free Energy, Blockchain, Anti-Gravity Propulsion, and even AI are extremely disruptive technologies and we should stop buying in to the fear-mongering and start understanding and developing them ourselves!
Nuclear Physics
Before I discuss Cold Fusion, let's first get an overview of Nuclear Physics.
Thus this video serves as a crash course in Nuclear Physics.
Note #1: This video mainly covers the general mainstream definitions of nuclear physics in order to get a better idea of the context of the physics that might entail #FreeEnergy.
Note #2: I will be mainly reviewing the government controlled Wikipedia to give a "mainstream" narrative on Nuclear Physics to get an overview, but if you find any point questionable please let me know as it will just further help uncover the true science being hidden from us!
Note #3: A Top Secret MES Life Hack is that the best way to self-learn is by teaching others. # ThisIsWhyIStartedMES
Now let's take a look at the mainstream view of some of the basics of Nuclear Physics.
https://en.wikipedia.org/wiki/Atom
Retrieved: 9 September 2017
Archive: https://archive.is/t1371
Atom
An atom is the smallest constituent unit of ordinary matter that has the properties of a chemical element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are very small; typical sizes are around 100 picometers (a ten-billionth of a meter, in the short scale).
Atoms are small enough that attempting to predict their behavior using classical physics – as if they were billiard balls, for example – gives noticeably incorrect predictions due to quantum effects. Through the development of physics, atomic models have incorporated quantum principles to better explain and predict the behavior.
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and typically a similar number of neutrons. Protons and neutrons are called nucleons. More than 99.94% of an atom's mass is in the nucleus. The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. If the number of protons and electrons are equal, that atom is electrically neutral. If an atom has more or fewer electrons than protons, then it has an overall negative or positive charge, respectively, and it is called an ion.
The electrons of an atom are attracted to the protons in an atomic nucleus by this electromagnetic force. The protons and neutrons in the nucleus are attracted to each other by a different force, the nuclear force, which is usually stronger than the electromagnetic force repelling the positively charged protons from one another. Under certain circumstances, the repelling electromagnetic force becomes stronger than the nuclear force, and nucleons can be ejected from the nucleus, leaving behind a different element: nuclear decay resulting in nuclear transmutation.
The number of protons in the nucleus defines to what chemical element the atom belongs: for example, all copper atoms contain 29 protons. The number of neutrons defines the isotope of the element. The number of electrons influences the magnetic properties of an atom. Atoms can attach to one or more other atoms by chemical bonds to form chemical compounds such as molecules. The ability of atoms to associate and dissociate is responsible for most of the physical changes observed in nature and is the subject of the discipline of chemistry.
An illustration of the helium atom, depicting the nucleus (pink) and the electron cloud distribution (black). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. The black bar is one angstrom (10-10 m or 100 pm).
https://en.wikipedia.org/wiki/Matter
Retrieved: 12 September 2017
Archive: https://archive.is/BXbja
Matter
In the classical physics observed in everyday life, matter is any substance that has mass and takes up space by having volume. This includes atoms and anything made up of these, but not other energy phenomena or waves such as light or sound.[1][2] More generally, however, in (modern) physics, matter is not a fundamental concept because a universal definition of it is elusive; for example, the elementary constituents of atoms may be point particles, each having no volume individually.
All the everyday objects that we can bump into, touch or squeeze are ultimately composed of atoms. This ordinary atomic matter is in turn made up of interacting subatomic particles—usually a nucleus of protons and neutrons, and a cloud of orbiting electrons.[3][4] Typically, science considers these composite particles matter because they have both rest mass and volume. By contrast, massless particles, such as photons, are not considered matter, because they have neither rest mass nor volume. However, not all particles with rest mass have a classical volume, since fundamental particles such as quarks and leptons (sometimes equated with matter) are considered "point particles" with no effective size or volume. Nevertheless, quarks and leptons together make up "ordinary matter", and their interactions contribute to the effective volume of the composite particles that make up ordinary matter.
Matter exists in states (or phases): the classical solid, liquid, and gas; as well as the more exotic plasma, Bose–Einstein condensates, fermionic condensates, and quark–gluon plasma.[5]
For much of the history of the natural sciences people have contemplated the exact nature of matter. The idea that matter was built of discrete building blocks, the so-called particulate theory of matter, was first put forward by the Greek philosophers Leucippus (~490 BC) and Democritus (~470–380 BC).[6]



Matter is usually classified into three classical states, with plasma sometimes added as a fourth state. From top to bottom: quartz (solid),water (liquid), nitrogen dioxide (gas), and a plasma globe (plasma).
…
Other types
Ordinary matter, in the quarks and leptons definition, constitutes about 4% of the energy of the observable universe. The remaining energy is theorized to be due to exotic forms, of which 23% is dark matter[48][49] and 73% is dark energy.[50][51]
Pie chart showing the fractions of energy in the universe contributed by different sources. Ordinary matter is divided into luminous matter (the stars and luminous gases and 0.005% radiation) and nonluminous matter (intergalactic gas and about 0.1% neutrinos and 0.04% supermassive black holes). Ordinary matter is uncommon. Modeled after Ostriker and Steinhardt.[47] For more information, see NASA.
Dark matter
Main articles: Dark matter, Lambda-CDM model, and WIMPs
See also: Galaxy formation and evolution and Dark matter haloIn astrophysics and cosmology, dark matter is matter of unknown composition that does not emit or reflect enough electromagnetic radiation to be observed directly, but whose presence can be inferred from gravitational effects on visible matter.[55][56]Observational evidence of the early universe and the big bang theory require that this matter have energy and mass, but is not composed ordinary baryons (protons and neutrons).
Dark energy
Main article: Dark energy
See also: Big bang § Dark energyIn cosmology, dark energy is the name given to source of the repelling influence that is accelerating the rate of expansion of the universe. Its precise nature is currently a mystery, although its effects can reasonably be modeled by assigning matter-like properties such as energy density and pressure to the vacuum itself.[58][59]
Fully 70% of the matter density in the universe appears to be in the form of dark energy. Twenty-six percent is dark matter. Only 4% is ordinary matter. So less than 1 part in 20 is made out of matter we have observed experimentally or described in the standard model of particle physics. Of the other 96%, apart from the properties just mentioned, we know absolutely nothing.
—Lee Smolin: The Trouble with Physics, p. 16Exotic matter
Main article: Exotic matterExotic matter is a concept of particle physics, which may include dark matter and dark energy but goes further to include any hypothetical material that violates one or more of the properties of known forms of matter. Some such materials might possess hypothetical properties like negative mass.
https://en.wikipedia.org/wiki/Chemical_element
Retrieved: 9 September 2017
Archive: https://archive.is/alYIt
Chemical element
A chemical element or element is a species of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, or Z).[1] There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up Earth, while oxygen is the most common element in the Earth's crust.[2]
Chemical elements constitute all of the ordinary matter of the universe. However astronomical observations suggest that ordinary observable matter makes up only about 15% of the matter in the universe: the remainder is dark matter; the composition of this is unknown, but it is not composed of chemical elements.[3]
https://en.wikipedia.org/wiki/Ion
Retrieved: 9 September 2017
Archive: https://archive.is/zekxV
Ion
An ion (/'a??n, -?n/)[1] is an atom, or a molecule, in which the total number of electrons is not equal to the total number of protons, giving the atom or molecule a net positive or negative electrical charge. An atom, or molecule, with a net positive charge is a cation. An atom, or molecule, with a net negative charge is an anion. Because of their opposite electric charges, cations and anions attract each other and readily form ionic compounds, such as salts.
Ions can be created by chemical means, such as the dissolution of a salt into water, or by physical means, such as passing a direct current through a conducting solution, which will dissolve the anode via ionization .
https://en.wikipedia.org/wiki/Plasma_(physics)
Retrieved: 9 September 2017
Archive: https://archive.is/imqII
Plasma (physics)
Plasma (from Ancient Greek p??sµa, meaning "moldable substance"[1]or "jelly")[2] is one of the four fundamental states of matter, while the others are solid, liquid, and gas. Unlike these three states of matter, plasma does not naturally exist on the Earth under normal surface conditions, and can only be artificially generated from neutral gases.[3]The term was first introduced by chemist Irving Langmuir[2] in the 1920s.[4]
Plasma and ionised gases have unique properties and display behaviors unlike those of the other states, although the true technical transition between the two is mostly a matter of nomenclature[2] and subject to interpretation.[5] It can simply be considered as a gaseous mixture of negatively charged electrons and highly charged positive ions, being created by heating a gas or by subjecting gas to a strong electromagnetic field. However, true plasma production is from the distinct separation of these ions and electrons that produces an electric field, which in turn, produces electric currents and magnetic fields.
…
Lightning is an example of plasma present at Earth's surface. Typically, lightning discharges 30,000 amperes at up to 100 million volts, and emits light, radio waves, X-rays and even gamma rays.[24] Plasma temperatures in lightning can approach 28,000 K (28,000 °C; 50,000 °F) and electron densities may exceed 1024 m-3.
https://en.wikipedia.org/wiki/Atomic_nucleus
Retrieved: 9 September 2017
Archive: https://archive.is/imne4
Atomic nucleus
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron in 1932, models for a nucleus composed of protons and neutrons were quickly developed by Dmitri Ivanenko[1] and Werner Heisenberg.[2][3][4][5][6] Almost all of the mass of an atom is located in the nucleus, with a very small contribution from the electron cloud. Protons and neutrons are bound together to form a nucleus by the nuclear force.
The diameter of the nucleus is in the range of 1.75 fm (1.75×10-15 m) for hydrogen (the diameter of a single proton)[7] to about 15 fm for the heaviest atoms, such as uranium. These dimensions are much smaller than the diameter of the atom itself (nucleus + electron cloud), by a factor of about 23,000 (uranium) to about 145,000 (hydrogen).[citation needed]
Retrieved: 9 September 2017
Archive: https://archive.is/qBHzr
Femtometre
The femtometre (American spelling femtometer, symbol fm[1][2][3] derived from the Danish and Norwegian word femten, "fifteen"+Ancient Greek: µ?t???, metr?n, "unit of measurement") is an SI unit of length equal to 10-15 metres, which means a quadrillionth of one. This distance can also be called a fermi and was so named in honour of physicist Enrico Fermi, as it is a typical length-scale of nuclear physics.
The branch of physics concerned with the study and understanding of the atomic nucleus, including its composition and the forces which bind it together, is called nuclear physics.
…
A model of the atomic nucleus showing it as a compact bundle of the two types of nucleons: protons (red) and neutrons (blue). In this diagram, protons and neutrons look like little balls stuck together, but an actual nucleus (as understood by modern nuclear physics) cannot be explained like this, but only by using quantum mechanics. In a nucleus which occupies a certain energy level (for example, the ground state), each nucleon can be said to occupy a range of locations.
…
A figurative depiction of the helium-4 atom with the electron cloud in shades of gray. In the nucleus, the two protons and two neutrons are depicted in red and blue. This depiction shows the particles as separate, whereas in an actual helium atom, the protons are superimposed in space and most likely found at the very center of the nucleus, and the same is true of the two neutrons. Thus, all four particles are most likely found in exactly the same space, at the central point. Classical images of separate particles fail to model known charge distributions in very small nuclei. A more accurate image is that the spatial distribution of nucleons in a helium nucleus is much closer to the helium electron cloud shown here, although on a far smaller scale, than to the fanciful nucleus image.
https://en.wikipedia.org/wiki/Atomic_orbital
Retrieved: 9 September 2017
Archive: https://archive.is/LM4MU
Atomic orbital
In quantum mechanics, an atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom.[1] This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The term, atomic orbital, may also refer to the physical region or space where the electron can be calculated to be present, as defined by the particular mathematical form of the orbital.[2]
…
Atomic orbitals are the basic building blocks of the atomic orbital model (alternatively known as the electron cloud or wave mechanics model), a modern framework for visualizing the submicroscopic behavior of electrons in matter.
…
https://en.wikipedia.org/wiki/Heat_map
Retrieved: 9 September 2017
Archive: https://archive.is/1qZXj
Heat map
A heat map (or heatmap) is a graphical representation of data where the individual values contained in a matrix are represented as colors. The term 'heat map' was originally coined and trademarked by software designer Cormac Kinney in 1991, to describe a 2D display depicting financial market information,[1] though similar plots such as shading matrices have existed for over a century.[2]
https://en.wikipedia.org/wiki/Quantum
Retrieved: 11 September 2017
Archive: https://archive.is/8I77d
Quantum
In physics, a quantum (plural: quanta) is the minimum amount of any physical entity involved in an interaction. The fundamental notion that a physical property may be "quantized" is referred to as "the hypothesis of quantization".[1] This means that the magnitude of the physical property can take on only certain discrete values.
For example, a photon is a single quantum of light (or of any other form of electromagnetic radiation), and can be referred to as a "light quantum". Similarly, the energy of an electron bound within an atom is also quantized, and thus can only exist in certain discrete values. The fact that electrons can only exist at discrete energy levels in an atom causes atoms to be stable, and hence matter in general is stable.
Quantization is one of the foundations of the much broader physics of quantum mechanics. Quantization of the energy and its influence on how energy and matter interact (quantum electrodynamics) is part of the fundamental framework for understanding and describing nature.
Etymology and discovery
The word quantum comes from the Latin quantus, meaning "how great". "Quanta", short for "quanta of electricity" (electrons), was used in a 1902 article on the photoelectric effect by Philipp Lenard, who credited Hermann von Helmholtz for using the word in the area of electricity. However, the word quantum in general was well known before 1900.[2]
https://en.wikipedia.org/wiki/Quantum_mechanics
Retrieved: 9 September 2017
Archive: https://archive.is/zMWP9
Quantum mechanics
Quantum mechanics (QM; also known as quantum physics or quantum theory), including quantum field theory, is a branch of physics which is the fundamental theory of nature at the small scales and energy levels of atoms and subatomic particles.[2]
Classical physics (the physics existing before quantum mechanics) derives from quantum mechanics as an approximation valid only at large (macroscopic[3]) scales. Quantum mechanics differs from classical physics in that: energy, momentum and other quantities are often restricted to discrete values (quantization), objects have characteristics of both particles and waves (i.e. wave-particle duality), and there are limits to the precision with which quantities can be known (uncertainty principle).[note 1]
Quantum mechanics gradually arose from Max Planck's solution in 1900 to the black-body radiation problem, and Albert Einstein's 1905 paper which offered a quantum-based theory to explain the photoelectric effect. Early quantum theory was profoundly re-conceived in the mid-1920s by Erwin Schrodinger, Werner Heisenberg, Max Born and others. The modern theory is formulated in various specially developed mathematical formalisms. In one of them, a mathematical function, the wave function, provides information about the probability amplitude of position, momentum, and other physical properties of a particle.
Wavefunctions of the electron in a hydrogen atom at different energy levels. Quantum mechanics cannot predict the exact location of a particle in space, only the probability of finding it at different locations.[1] The brighter areas represent a higher probability of finding the electron.
https://en.wikipedia.org/wiki/Wave%E2%80%93particle_duality
Retrieved: 13 September 2017
Archive: https://archive.is/UWC70
Wave-particle duality
Wave–particle duality is the concept in quantum mechanics that every particle or quantic entity may be partly described in terms not only of particles, but also of waves. It expresses the inability of the classical concepts "particle" or "wave" to fully describe the behavior of quantum-scale objects. As Albert Einstein wrote:[1]
It seems as though we must use sometimes the one theory and sometimes the other, while at times we may use either. We are faced with a new kind of difficulty. We have two contradictory pictures of reality; separately neither of them fully explains the phenomena of light, but together they do.
Through the work of Max Planck, Einstein, Louis de Broglie, Arthur Compton, Niels Bohr and many others, current scientific theory holds that all particles also have a wave nature (and vice versa).[2] This phenomenon has been verified not only for elementary particles, but also for compound particles like atoms and even molecules. For macroscopic particles, because of their extremely short wavelengths, wave properties usually cannot be detected.[3]
…
Animation showing the wave-particle duality with a double slit experiment and effect of an observer. Increase size to see explanations in the video itself. See also quiz based on this animation.
https://commons.wikimedia.org/w/index.php?title=File%3AWave-particle_duality.ogv
Retrieved: 13 September 2017
Archive: https://archive.is/UWC70
Very interesting! Appears that the act of "observing" affects the observations themselves! # MindBlown
https://en.wikipedia.org/wiki/Subatomic_particle
Retrieved: 9 September 2017
Archive: https://archive.is/Texdf
Subatomic particle
In the physical sciences, subatomic particles are particles much smaller than atoms.[1] There are two types of subatomic particles: elementary particles, which according to current theories are not made of other particles; and composite particles.[2] Particle physics and nuclear physics study these particles and how they interact.[3]
Standard model of elementary particles: the 12 fundamental fermions and 4 fundamental bosons. Brown loops indicate which bosons (red) couple to which fermions (purple and green). Please note that the masses of certain particles are subject to periodic reevaluation by the scientific community. The values currently reflected in this graphic are as of 2008 and may have been adjusted since. For the latest consensus, please visit the Particle Data Group website linked below.
https://en.wikipedia.org/wiki/Fundamental_interaction
Retrieved: 10 September 2017
Archive: https://archive.is/HAMek
Fundamental interaction
In physics, the fundamental interactions, also known as fundamental forces, are the interactions that do not appear to be reducible to more basic interactions. There are four fundamental interactions known to exist: the gravitational and electromagnetic interactions, which produce significant long-range forces whose effects can be seen directly in everyday life, and the strong, and weak interactions, which produce forces at minuscule, subatomic distances and govern nuclear interactions. Some scientists[who?]speculate that a fifth force might exist but if so, it is not widely accepted nor proven.
Each of the known fundamental interactions can be described mathematically as a field. The gravitational force is attributed to the curvature of spacetime, described by Einstein's general theory of relativity. The other three are discrete quantum fields, and their interactions are mediated by elementary particles described by the Standard Model of particle physics.
Within the Standard Model, the strong interaction is carried by a particle called the gluon, and is responsible for the binding of quarks together to form hadrons, such as protons and neutrons. As a residual effect, it creates the nuclear force that binds the latter particles to form atomic nuclei. The weak interaction is carried by particles called W and Z bosons, and also acts on the nucleus of atoms, mediating radioactive decay. The electromagnetic force, carried by the photon, creates electric and magnetic fields, which are responsible for chemical bonding and electromagnetic waves, including visible light, and forms the basis for electrical technology. Although the electromagnetic force is far stronger than gravity, it tends to cancel itself out within large objects, so over the largest distances (on the scale of planets and galaxies), gravity tends to be the dominant force.
All four fundamental forces are believed to be related, and to unite into a single force at high energies on a minuscule scale, the Planck scale, but particle accelerators cannot produce the enormous energies required to experimentally probe this. Efforts to devise a common theoretical framework that would explain the relation between the forces are perhaps the greatest goal of theoretical physicists today. The weak and electromagnetic forces have already been unified with the electroweak theory of Sheldon Glashow, Abdus Salam, and Steven Weinberg for which they received the 1979 Nobel Prize in physics. Progress is currently being made in uniting the electroweak and strong fields within a Grand Unified Theory (GUT). A bigger challenge is to find a way to quantize the gravitational field, resulting in a theory of quantum gravity (QG) which would unite gravity in a common theoretical framework with the other three forces. Some theories, notably string theory, seek both QG and GUT within one framework, unifying all four fundamental interactions along with mass generation within a theory of everything (ToE).
https://en.wikipedia.org/wiki/Gravity
Retrieved: 10 September 2017
Archive: https://archive.is/yUZBk
Gravity
Gravity, or gravitation, is a natural phenomenon by which all things with mass are brought toward (or gravitate toward) one another, including planets, stars and galaxies, and other physical objects. Since energy and mass are equivalent, all forms of energy (including light) cause gravitation and are under the influence of it. On Earth, gravity gives weight to physical objects, and causes the ocean tides. The gravitational attraction of the original gaseous matter present in the Universe caused it to begin coalescing, forming stars – and for the stars to group together into galaxies – so gravity is responsible for many of the large scale structures in the Universe. Gravity has an infinite range, although its effects become increasingly weaker on farther objects.
Gravity is most accurately described by the general theory of relativity (proposed by Albert Einstein in 1915) which describes gravity not as a force, but as a consequence of the curvature of spacetime caused by the uneven distribution of mass. The most extreme example of this curvature of spacetime is a black hole, from which nothing can escape once past its event horizon, not even light.[1] However, for most applications, gravity is well approximated by Newton's law of universal gravitation, which describes gravity as a force which causes any two bodies to be attracted to each other, with the force proportional to the product of their masses and inversely proportional to the square of the distance between them.
Gravity is the weakest of the four fundamental forces of physics, approximately 1038 times weaker than the strong force, 1036 times weaker than the electromagnetic force and 1029 times weaker than the weak force. As a consequence, it has no significant influence at the level of subatomic particles.[2] In contrast, it is the dominant force at the macroscopic scale, and is the cause of the formation, shape and trajectory (orbit) of astronomical bodies. For example, gravity causes the Earth and the other planets to orbit the Sun, it also causes the Moon to orbit the Earth, and causes the formation of tides, the formation and evolution of the Solar System, stars and galaxies.
The earliest instance of gravity in the Universe, possibly in the form of quantum gravity, supergravity or a gravitational singularity, along with ordinary space and time, developed during the Planck epoch (up to 10-43 seconds after the birth of the Universe), possibly from a primeval state, such as a false vacuum, quantum vacuum or virtual particle, in a currently unknown manner.[3] Attempts to develop a theory of gravity consistent with quantum mechanics, a quantum gravity theory, which would allow gravity to be united in a common mathematical framework (a theory of everything) with the other three forces of physics, are a current area of research.
Two-dimensional analogy of spacetime distortion generated by the mass of an object. Matter changes the geometry of spacetime, this (curved) geometry being interpreted as gravity. White lines do not represent the curvature of space but instead represent the coordinate system imposed on the curved spacetime, which would be rectilinear in a flat spacetime.
https://en.wikipedia.org/wiki/Electromagnetism
Retrieved: 10 September 2017
Archive: https://archive.is/fxDf8
Electromagnetism
Electromagnetism is a branch of physics involving the study of the electromagnetic force, a type of physical interaction that occurs between electrically charged particles. The electromagnetic force usually exhibits electromagnetic fields such as electric fields, magnetic fields, and light and is one of the four fundamental interactions (commonly called forces) in nature. The other three fundamental interactions are the strong interaction, the weak interaction and gravitation.[1]
The word electromagnetism is a compound form of two Greek terms, ??e?t??? elektron, "amber", and µa???t?? ????? magnetis lithos,[2] which means "?agnesian stone",[3] a type of iron ore. Electromagnetic phenomena are defined in terms of the electromagnetic force, sometimes called the Lorentz force, which includes both electricity and magnetism as different manifestations of the same phenomenon.
The electromagnetic force plays a major role in determining the internal properties of most objects encountered in daily life. Ordinary matter takes its form as a result of intermolecular forces between individual atoms and molecules in matter, and is a manifestation of the electromagnetic force. Electrons are bound by the electromagnetic force to atomic nuclei, and their orbital shapes and their influence on nearby atoms with their electrons is described by quantum mechanics. The electromagnetic force governs the processes involved in chemistry, which arise from interactions between the electrons of neighboring atoms.
There are numerous mathematical descriptions of the electromagnetic field. In classical electrodynamics, electric fields are described as electric potential and electric current. In Faraday's law, magnetic fields are associated with electromagnetic induction and magnetism, and Maxwell's equations describe how electric and magnetic fields are generated and altered by each other and by charges and currents.
The theoretical implications of electromagnetism, particularly the establishment of the speed of light based on properties of the "medium" of propagation (permeability and permittivity), led to the development of special relativity by Albert Einstein in 1905.
Although electromagnetism is considered one of the four fundamental forces, at high energy the weak force and electromagnetic force are unified as a single electroweak force. In the history of the universe, during the quark epoch the unified force broke into the two separate forces as the universe cooled.
Lightning is an electrostatic discharge that travels between two charged regions.
https://en.wikipedia.org/wiki/Strong_interaction
Retrieved: 11 September 2017
Archive: https://archive.is/Ku4gX
Strong interaction
In particle physics, the strong interaction is the mechanism responsible for the strong nuclear force (also called the strong force or nuclear strong force), and is one of the four known fundamental interactions, with the others being electromagnetism, the weak interaction and gravitation. At the range of 10-15 m (1 femtometer), the strong force is approximately 137 times as strong as electromagnetism, a million times as strong as the weak interaction and 1038 times as strong as gravitation.[1] The strong nuclear force holds most ordinary matter together because it confines quarks into hadron particles such as the proton and neutron. In addition, the strong force binds neutrons and protons to create atomic nuclei. Most of the mass of a common proton or neutron is the result of the strong force field energy; the individual quarks provide only about 1% of the mass of a proton.
The strong interaction is observable at two ranges: on a larger scale (about 1 to 3 fm), it is the force that binds protons and neutrons (nucleons) together to form the nucleus of an atom. On the smaller scale (less than about 0.8 fm, the radius of a nucleon), it is the force (carried by gluons) that holds quarks together to form protons, neutrons, and other hadron particles. In the latter context, it is often known as the color force. The strong force inherently has such a high strength that hadrons bound by the strong force can produce new massive particles. Thus, if hadrons are struck by high-energy particles, they give rise to new hadrons instead of emitting freely moving radiation (gluons). This property of the strong force is called color confinement, and it prevents the free "emission" of the strong force: instead, in practice, jets of massive particles are produced.
In the context of binding protons and neutrons together to form atomic nuclei, the strong interaction is called the nuclear force (or residual strong force). In this case, it is the residuum of the strong interaction between the quarks that make up the protons and neutrons. As such, the residual strong interaction obeys a quite different distance-dependent behavior between nucleons, from when it is acting to bind quarks within nucleons. The binding energy that is partly released on the breakup of a nucleus is related to the residual strong force and is harnessed as fission energy in nuclear power and fission-type nuclear weapons.[2][3]
The strong interaction is mediated by the exchange of massless particles called gluons that act between quarks, antiquarks, and other gluons. Gluons are thought to interact with quarks and other gluons by way of a type of charge called color charge. Color charge is analogous to electromagnetic charge, but it comes in three types (±red, ±green, ±blue) rather than one, which results in a different type of force, with different rules of behavior. These rules are detailed in the theory of quantum chromodynamics (QCD), which is the theory of quark-gluon interactions.
After the Big Bang and during the electroweak epoch of the universe, the electroweak force separated from the strong force. A Grand Unified Theory is hypothesized to have existed to describe this, but no such theory has yet been successfully formulated, and the unification remains an unsolved problem in physics.
The nucleus of a helium atom. The two protons have the same charge, but still stay together due to the residual nuclear force
https://en.wikipedia.org/wiki/Nuclear_force
Retrieved: 11 September 2017
Archive: https://archive.is/5miuP
Nuclear force
The nuclear force (or nucleon–nucleon interaction or residual strong force) is a force that acts between the protons and neutrons of atoms. Neutrons and protons, both nucleons, are affected by the nuclear force almost identically. Since protons have charge +1 e, they experience an electric force that tends to push them apart, but at short range the attractive nuclear force is strong enough to overcome the electromagnetic force. The nuclear force binds nucleons into atomic nuclei.
The nuclear force is powerfully attractive between nucleons at distances of about 1 femtometre (fm, or 1.0 × 10-15 metres), but it rapidly decreases to insignificance at distances beyond about 2.5 fm. At distances less than 0.7 fm, the nuclear force becomes repulsive. This repulsive component is responsible for the physical size of nuclei, since the nucleons can come no closer than the force allows. By comparison, the size of an atom, measured in angstroms (Å, or 1.0 × 10-10 m), is five orders of magnitude larger. The nuclear force is not simple, however, since it depends on the nucleon spins, has a tensor component, and may depend on the relative momentum of the nucleons.[2] The nuclear force is not one of the fundamental forces of nature.
The nuclear force plays an essential role in storing energy that is used in nuclear power and nuclear weapons. Work (energy) is required to bring charged protons together against their electric repulsion. This energy is stored when the protons and neutrons are bound together by the nuclear force to form a nucleus. The mass of a nucleus is less than the sum total of the individual masses of the protons and neutrons. The difference in masses is known as the mass defect, which can be expressed as an energy equivalent. Energy is released when a heavy nucleus breaks apart into two or more lighter nuclei. This energy is the electromagnetic potential energy that is released when the nuclear force no longer holds the charged nuclear fragments together.[3][4]
A quantitative description of the nuclear force relies on equations that are partly empirical. These equations model the internucleon potential energies, or potentials. (Generally, forces within a system of particles can be more simply modeled by describing the system's potential energy; the negative gradient of a potential is equal to the vector force.) The constants for the equations are phenomenological, that is, determined by fitting the equations to experimental data.
…
Corresponding potential energy (in units of MeV) of two nucleons as a function of distance as computed from the Reid potential. The potential well is a minimum at a distance of about 0.8 fm. With this potential nucleons can become bound with a negative "binding energy."
https://en.wikipedia.org/wiki/Weak_interaction
Retrieved: 11 September 2017
Archive: https://archive.is/1Lk7F
Weak interaction
In particle physics, the weak interaction (the weak force or weak nuclear force) is the mechanism of interaction between sub-atomic particles that causes radioactive decay and thus plays an essential role in nuclear fission. The theory of the weak interaction is sometimes called quantum flavourdynamics (QFD), in analogy with the terms QCD dealing with the strong interaction and QED dealing with the electromagnetic force. However the term QFD is rarely used because the weak force is best understood in terms of electro-weak theory (EWT).[1]
The weak interaction takes place only at very small, sub-atomic distances, less than the diameter of a proton. It is one of the four known fundamental interactions of nature, alongside the strong interaction, electromagnetism, and gravitation.
The radioactive beta decay is due to the weak interaction, which transforms a neutron into: a proton, an electron, and an electron antineutrino.
https://en.wikipedia.org/wiki/Electron_neutrino
Retrieved: 11 September 2017
Archive: https://archive.is/AMCDg
Electron neutrino
The electron neutrino (?e) is a subatomic lepton elementary particle which has no net electric charge. Together with the electron it forms the first generation of leptons, hence the name electron neutrino. It was first hypothesized by Wolfgang Pauli in 1930, to account for missing momentum and missing energy in beta decay, and was discovered in 1956 by a team led by Clyde Cowan and Frederick Reines (see Cowan–Reines neutrino experiment).[1]
Proposal
In the early 1900s, theories predicted that the electrons resulting from beta decay should have been emitted at a specific energy. However, in 1914, James Chadwick showed that electrons were instead emitted in a continuous spectrum.[1]
In 1930, Wolfgang Pauli theorized that an undetected particle was carrying away the observed difference between the energy, momentum, and angular momentum of the initial and final particles.[nb 1][2]
https://en.wikipedia.org/wiki/Particle_accelerator
Retrieved: 9 September 2017
Archive: https://archive.is/GBUud
Particle accelerator
A particle accelerator is a machine that uses electromagnetic fields to propel charged particles to nearly light speed and to contain them in well-defined beams.[1]
Large accelerators are used in particle physics as colliders (e.g., the LHC at CERN, KEKB at KEK in Japan, RHIC at Brookhaven National Laboratory, and Tevatron at Fermilab), or as synchrotron light sources for the study of condensed matter physics. Smaller particle accelerators are used in a wide variety of applications, including particle therapy for oncological purposes, radioisotope production for medical diagnostics, ion implanters for manufacture of semiconductors, and accelerator mass spectrometers for measurements of rare isotopes such as radiocarbon. There are currently more than 30,000 accelerators in operation around the world.[2]
There are two basic classes of accelerators: electrostatic and electrodynamic (or electromagnetic) accelerators. [3] Electrostatic accelerators use static electric fields to accelerate particles. The most common types are the Cockcroft–Walton generator and the Van de Graaff generator. A small-scale example of this class is the cathode ray tube in an ordinary old television set. The achievable kinetic energy for particles in these devices is determined by the accelerating voltage, which is limited by electrical breakdown. Electrodynamic or electromagnetic accelerators, on the other hand, use changing electromagnetic fields (either magnetic induction or oscillating radio frequency fields) to accelerate particles. Since in these types the particles can pass through the same accelerating field multiple times, the output energy is not limited by the strength of the accelerating field. This class, which was first developed in the 1920s, is the basis for most modern large-scale accelerators.
Rolf Widerøe, Gustav Ising, Leó Szilárd, Max Steenbeck, and Ernest Lawrence are considered pioneers of this field, conceiving and building the first operational linear particle accelerator,[4] the betatron, and the cyclotron.
Because colliders can give evidence of the structure of the subatomic world, accelerators were commonly referred to as atom smashers in the 20th century.[5] Despite the fact that most accelerators (but not ion facilities) actually propel subatomic particles, the term persists in popular usage when referring to particle accelerators in general.[6][7][8]
Sketch of an electrostatic Van de Graaff accelerator
Sketch of the Ising/Widerøe linear accelerator concept, employing oscillating fields (1928)
…
Aerial photo of the Tevatron at Fermilab, which resembles a figure eight. The main accelerator is the ring above; the one below (about half the diameter, despite appearances) is for preliminary acceleration, beam cooling and storage, etc.
…
Higher energies
At present the highest energy accelerators are all circular colliders, but both hadron accelerators and electron accelerators are running into limits. Higher energy hadron and ion cyclic accelerators will require accelerator tunnels of larger physical size due to the increased beam rigidity.
A Livingston chart depicting progress in collision energy through 2010. The LHC is the largest collision energy to date, but also represents the first break in the log-linear trend.
…
Black hole production and public safety concerns
See also: Safety of high energy particle collision experimentsIn the future, the possibility of black hole production at the highest energy accelerators may arise if certain predictions of superstring theory are accurate.[27][28] This and other possibilities have led to public safety concerns that have been widely reported in connection with the LHC, which began operation in 2008. The various possible dangerous scenarios have been assessed as presenting "no conceivable danger" in the latest risk assessment produced by the LHC Safety Assessment Group.[29]
https://en.wikipedia.org/wiki/Elementary_particle
Retrieved: 14 September 2017
Archive: https://archive.is/PjD45
Elementary particle
In particle physics, an elementary particle or fundamental particle is a particle whose substructure is unknown; thus, it is unknown whether it is composed of other particles.[1] Known elementary particles include the fundamental fermions (quarks, leptons, antiquarks, and antileptons), which generally are "matter particles" and "antimatter particles", as well as the fundamental bosons (gauge bosons and the Higgs boson), which generally are "force particles" that mediate interactions among fermions.[1] A particle containing two or more elementary particles is a composite particle.
Everyday matter is composed of atoms, once presumed to be matter's elementary particles—atom meaning "unable to cut" in Greek—although the atom's existence remained controversial until about 1910, as some leading physicists regarded molecules as mathematical illusions, and matter as ultimately composed of energy.[1][2] Soon, subatomic constituents of the atom were identified. As the 1930s opened, the electron and the proton had been observed, along with the photon, the particle of electromagnetic radiation.[1] At that time, the recent advent of quantum mechanics was radically altering the conception of particles, as a single particle could seemingly span a field as would a wave, a paradox still eluding satisfactory explanation.[3][4][5]
Via quantum theory, protons and neutrons were found to contain quarks—up quarks and down quarks—now considered elementary particles.[1] And within a molecule, the electron's three degrees of freedom (charge, spin, orbital) can separate via the wavefunction into three quasiparticles (holon, spinon, orbiton).[6] Yet a free electron—which is not orbiting an atomic nucleus and lacks orbital motion—appears unsplittable and remains regarded as an elementary particle.[6]
Around 1980, an elementary particle's status as indeed elementary—an ultimate constituent of substance—was mostly discarded for a more practical outlook,[1] embodied in particle physics' Standard Model, what's known as science's most experimentally successful theory.[5][7] Many elaborations upon and theories beyond the Standard Model, including the popular supersymmetry, double the number of elementary particles by hypothesizing that each known particle associates with a "shadow" partner far more massive,[8][9] although all such superpartners remain undiscovered.[7][10] Meanwhile, an elementary boson mediating gravitation—the graviton—remains hypothetical.[1]
…
Notes:
- The antielectron (e+) is traditionally called positron
- The known force carrier bosons all have spin = 1 and are therefore vector bosons. The hypothetical graviton has spin = 2 and is a tensor boson; if it is a gauge boson as well is unknown.
https://en.wikipedia.org/wiki/Spin_(physics)
Retrieved: 11 September 2017
Archive: https://archive.is/80rgk
Spin (physics)
In quantum mechanics and particle physics, spin is an intrinsic form of angular momentum carried by elementary particles, composite particles (hadrons), and atomic nuclei.[1][2]
Spin is one of two types of angular momentum in quantum mechanics, the other being orbital angular momentum. The orbital angular momentum operator is the quantum-mechanical counterpart to the classical angular momentum of orbital revolution: it arises when a particle executes a rotating or twisting trajectory (such as when an electron orbits a nucleus).[3][4] The existence of spin angular momentum is inferred from experiments, such as the Stern–Gerlach experiment, in which particles are observed to possess angular momentum that cannot be accounted for by orbital angular momentum alone.[5]
In some ways, spin is like a vector quantity; it has a definite magnitude, and it has a "direction" (but quantization makes this "direction" different from the direction of an ordinary vector). All elementary particles of a given kind have the same magnitude of spin angular momentum, which is indicated by assigning the particle a spin quantum number.[2]
The SI unit of spin is the (N·m·s) or (kg·m2·s-1), just as with classical angular momentum.
…
Elementary particles which are thought of as carrying forces are all bosons with spin 1. They include the photon which carries the electromagnetic force, the gluon (strong force), and the W and Z bosons (weak force). The ability of bosons to occupy the same quantum state is used in the laser, which aligns many photons having the same quantum number (the same direction and frequency), superfluid liquid helium resulting from helium-4 atoms being bosons, and superconductivity where pairs of electrons (which individually are fermions) act as single composite bosons.
…
Since 2013, the Higgs boson with spin 0 has been considered proven to exist.[6] It is the first scalar elementary particle (spin 0) known to exist in nature.
Theoretical and experimental studies have shown[citation needed] that the spin possessed by elementary particles cannot be explained by postulating that they are made up of even smaller particles rotating about a common center of mass analogous to a classical electron radius; as far as can be determined at present, these elementary particles have no inner structure. The spin of an elementary particle is therefore seen as a truly intrinsic physical property, akin to the particle's electric charge and rest mass.
https://en.wikipedia.org/wiki/Hadron
Retrieved: 10 September 2017
Archive: https://archive.is/d5wQy
Hadron
In particle physics, a hadron /'hædr?n/ (listen) (Greek: ?d???, hadrós, "stout, thick") is a composite particle made of quarks held together by the strong force in a similar way as molecules are held together by the electromagnetic force.
Hadrons are categorized into two families: baryons, made of three quarks, and mesons, made of one quark and one antiquark. Protons and neutrons are examples of baryons; pions are an example of a meson. Hadrons containing more than three valence quarks (exotic hadrons) have been discovered in recent years.
https://en.wikipedia.org/wiki/Boson
Retrieved: 11 September 2017
Archive: https://archive.is/ciFy8
Boson
In quantum mechanics, a boson (/'bo?s?n/,[1] /'bo?z?n/[2]) is a particle that follows Bose–Einstein statistics. Bosons make up one of the two classes of particles, the other being fermions.[3]
Examples of bosons include fundamental particles such as photons, gluons, and W and Z bosons (the four force-carrying gauge bosons of the Standard Model), the recently discovered Higgs boson, and the hypothetical graviton of quantum gravity. Some composite particles are also bosons, such as mesons and stable nuclei of even mass number such as deuterium (with one proton and one neutron, mass number = 2), helium-4, or lead-208[Note 1]; as well as some quasiparticles (e.g. Cooper pairs, plasmons, and phonons).[4]:130
An important characteristic of bosons is that their statistics do not restrict the number of them that occupy the same quantum state. This property is exemplified by helium-4 when it is cooled to become a superfluid.[5] Unlike bosons, two identical fermions cannot occupy the same quantum space. Whereas the elementary particles that make up matter (i.e. leptons and quarks) are fermions, the elementary bosons are force carriers that function as the 'glue' holding matter together.[6]
…
All observed elementary particles are either fermions or bosons. The observed elementary bosons are all gauge bosons: photons, W and Z bosons, gluons, except the Higgs boson which is a scalar boson.
• Photons are the force carriers of the electromagnetic field.
• W and Z bosons are the force carriers which mediate the weak force.
• Gluons are the fundamental force carriers underlying the strong force.
• Higgs bosons give W and Z bosons mass via the Higgs mechanism. Their existence was confirmed by CERN on 14 March 2013.Finally, many approaches to quantum gravity postulate a force carrier for gravity, the graviton, which is a boson of spin plus or minus two.
https://en.wikipedia.org/wiki/Higgs_boson
Retrieved: 11 September 2017
Archive: https://archive.is/dlkzP
Higgs boson
The Higgs boson is an elementary particle in the Standard Model of particle physics. First suspected to exist in the 1960s, it is the quantum excitation of the Higgs field,[6][7] a fundamental field of crucial importance to particle physics theory.[7] Unlike other known fields such as the electromagnetic field, it has a non-zero constant value in vacuum. The question of the existence of the Higgs field became the last unverified part of the Standard Model of particle physics, and for several decades, was considered "the central problem in particle physics".[8][9]
The presence of the field, now confirmed by experimental investigation, explains why some fundamental particles have mass when, based on the symmetries controlling their interactions, they should be massless. It also resolves several other long-standing puzzles, such as the reason for the extremely short range of the weak force.
Although the Higgs field is non-zero everywhere and its effects ubiquitous, proving its existence was far from easy. In principle, it can be proved to exist by detecting its excitations, which manifest as Higgs particles (the Higgs boson), but these are extremely difficult to produce and to detect. The importance of this fundamental question led to a 40-year search, and the construction of one of the world's most expensive and complex experimental facilities to date, CERN's Large Hadron Collider,[10] in an attempt to create Higgs bosons and other particles for observation and study. On 4 July 2012, the discovery of a new particle with a mass between 125 and 127 GeV/c2 was announced; physicists suspected that it was the Higgs boson.[11][12][13] Since then, the particle has been shown to behave, interact, and decay in many of the ways predicted for Higgs particles by the Standard Model, as well as having even parity and zero spin,[4][5] two fundamental attributes of a Higgs boson. This also means it is the first elementary scalar particle discovered in nature.[14] More studies are needed to verify with higher precision that the discovered particle has properties matching those predicted for the Higgs boson by the Standard Model, or whether, as predicted by some theories, multiple Higgs bosons exist.[15]
The Higgs boson is named after Peter Higgs, one of six physicists who, in the 1964 PRL symmetry breaking papers, proposed the Higgs mechanism that suggested the existence of such a particle. On December 10, 2013, two of the physicists, Peter Higgs and François Englert, were awarded the Nobel Prize in Physics for their work and prediction (Englert's co-researcher Robert Brout had died in 2011 and the Nobel Prize is not ordinarily given posthumously).[16] Although Higgs's name has come to be associated with this theory, several researchers between about 1960 and 1972 independently developed different parts of it. In mainstream media the Higgs boson has often been called the "God particle", from a 1993 book on the topic;[17] the nickname is strongly disliked by many physicists, including Higgs, who regard it as sensationalistic.[18][19][20]
In the Standard Model, the Higgs particle is a boson with no spin, electric charge, or colour charge. It is also very unstable, decaying into other particles almost immediately. It is a quantum excitation of one of the four components of the Higgs field.
Candidate Higgs boson events from collisions between protons in the LHC. The top event in the CMS experiment shows a decay into two photons (dashed yellow lines and green towers). The lower event in the ATLAS experiment shows a decay into four muons (red tracks).[Note 1]
https://en.wikipedia.org/wiki/Electron
Retrieved: 9 September 2017
Archive: https://archive.is/5nwfjElectron
The electron is a subatomic particle, symbol e- or ß-, with a negative elementary electric charge.[8]Electrons belong to the first generation of the lepton particle family,[9] and are generally thought to be elementary particles because they have no known components or substructure.[1] The electron has a mass that is approximately 1/1836 that of the proton.[10] Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of a half-integer value, expressed in units of the reduced Planck constant, h. As it is a fermion, no two electrons can occupy the same quantum state, in accordance with the Pauli exclusion principle.[9]Like all elementary particles, electrons exhibit properties of both particles and waves: they can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer De Broglie wavelength for a given energy.
Hydrogen atom orbitals at different energy levels. The brighter areas are where one is most likely to find an electron at any given time.
https://en.wikipedia.org/wiki/Proton
Retrieved: 9 September 2017
Archive: https://archive.is/D1Akg
Proton
A proton is a subatomic particle, symbol p or p+, with a positive electric charge of +1e elementary charge and mass slightly less than that of a neutron. Protons and neutrons, each with masses of approximately one atomic mass unit, are collectively referred to as "nucleons".
One or more protons are present in the nucleus of every atom; they are a necessary part of the nucleus. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol Z). Since each element has a unique number of protons, each element has its own unique atomic number.
The word proton is Greek for "first", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. In previous years, Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by atomic collisions. Protons were therefore a candidate to be a fundamental particle, and hence a building block of nitrogen and all other heavier atomic nuclei.
In the modern Standard Model of particle physics, protons are hadrons, and like neutrons, the other nucleon (particles present in atomic nuclei), are composed of three quarks. Although protons were originally considered fundamental or elementary particles, they are now known to be composed of three valence quarks: two up quarks and one down quark. The rest masses of quarks contribute only about 1% of a proton's mass, however.[3] The remainder of a proton's mass is due to quantum chromodynamics binding energy, which includes the kinetic energy of the quarks and the energy of the gluon fields that bind the quarks together. Because protons are not fundamental particles, they possess a physical size, though not a definite one; the root mean square charge radius of a proton is about 0.84–0.87 fm or 0.84×10-15 to 0.87×10-15 m.[4][5]
At sufficiently low temperatures, free protons will bind to electrons. However, the character of such bound protons does not change, and they remain protons. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. The result is a protonated atom, which is a chemical compound of hydrogen. In vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. Such "free hydrogen atoms" tend to react chemically with many other types of atoms at sufficiently low energies. When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H2), which are the most common molecular component of molecular clouds in interstellar space.
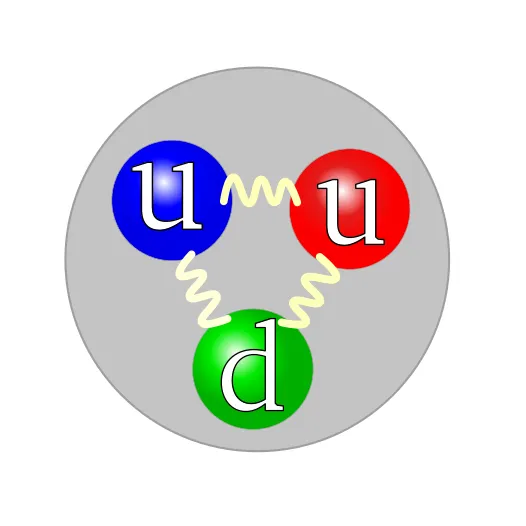
The quark structure of a proton. The color assignment of individual quarks is arbitrary, but all three colors must be present. Forces between quarks are mediated by gluons.
https://en.wikipedia.org/wiki/Unified_atomic_mass_unit
Retrieved: 9 September 2017
Archive: https://archive.is/hL2Xo
Unified atomic mass unit
The unified atomic mass unit (symbol: u) or dalton (symbol: Da) is a standard unit of mass that quantifies mass on an atomic or molecular scale (atomic mass). One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.[1] It is defined as one twelfth of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state and at rest,[2] and has a value of 1.660539040(20)×10-27 kg, or approximately 1.66 yoctograms.[3] The CIPM has categorised it as a non-SI unit accepted for use with the SI, and whose value in SI units must be obtained experimentally.[2]
The amu without the "unified" prefix is technically an obsolete unit based on oxygen, which was replaced in 1961. However, many sources still use the term "amu" but now define it in the same way as u (i.e., based on carbon-12).[4][5] In this sense, most uses of the terms "atomic mass units" and "amu" today actually refer to unified atomic mass unit. For standardization a specific atomic nucleus (carbon-12 vs. oxygen-16) had to be chosen because the average mass of a nucleon depends on the count of the nucleons in the atomic nucleus due to mass defect. This is also why the mass of a proton or neutron by itself is more than (and not equal to) 1 u.
https://en.wikipedia.org/wiki/Quark
Retrieved: 9 September 2017
Archive: https://archive.is/7JNRX
Quark
A quark (/'kw??rk/ or /'kw??rk/) is a type of elementary particle and a fundamental constituent of matter. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.[1] Due to a phenomenon known as color confinement, quarks are never directly observed or found in isolation; they can be found only within hadrons, such as baryons (of which protons and neutrons are examples) and mesons.[2][3] For this reason, much of what is known about quarks has been drawn from observations of the hadrons themselves.
Quarks have various intrinsic properties, including electric charge, mass, color charge, and spin. Quarks are the only elementary particles in the Standard Model of particle physics to experience all four fundamental interactions, also known as fundamental forces (electromagnetism, gravitation, strong interaction, and weak interaction), as well as the only known particles whose electric charges are not integer multiples of the elementary charge.
There are six types of quarks, known as flavors: up, down, strange, charm, top, and bottom.[4] Up and down quarks have the lowest masses of all quarks. The heavier quarks rapidly change into up and down quarks through a process of particle decay: the transformation from a higher mass state to a lower mass state. Because of this, up and down quarks are generally stable and the most common in the universe, whereas strange, charm, bottom, and top quarks can only be produced in high energy collisions (such as those involving cosmic rays and in particle accelerators). For every quark flavor there is a corresponding type of antiparticle, known as an antiquark, that differs from the quark only in that some of its properties have equal magnitude but opposite sign.
The quark model was independently proposed by physicists Murray Gell-Mann and George Zweig in 1964.[5]Quarks were introduced as parts of an ordering scheme for hadrons, and there was little evidence for their physical existence until deep inelastic scattering experiments at the Stanford Linear Accelerator Center in 1968.[6][7]Accelerator experiments have provided evidence for all six flavors. The top quark was the last to be discovered at Fermilab in 1995.[5]
A proton is composed of two up quarks, one down quark, and the gluons that mediate the forces "binding" them together. The color assignment of individual quarks is arbitrary, but all three colors must be present.
…
Six of the particles in the Standard Model are quarks (shown in purple). Each of the first three columns forms a generation of matter.
https://en.wikipedia.org/wiki/Color_charge
Retrieved: 9 September 2017
Archive: https://archive.is/WxulA
Color charge
Color charge is a property of quarks and gluons that is related to the particles' strong interactions in the theory of quantum chromodynamics (QCD).
The "color charge" of quarks and gluons is completely unrelated to the everyday meaning of color.[1] The term color and the labels red, green, blue became popular simply because of the loose analogy to the primary colors. (Richard Feynman referred to his colleagues as "idiot physicists" for choosing the confusing name.)
https://en.wikipedia.org/wiki/Gluon
Retrieved: 9 September 2017
Archive: https://archive.is/w7e3r
Gluon
A gluon /'glu??n/ is an elementary particle that acts as the exchange particle (or gauge boson) for the strong force between quarks. It is analogous to the exchange of photons in the electromagnetic force between two charged particles.[6] In layman's terms, they "glue" quarks together, forming protons and neutrons.
In technical terms, gluons are vector gauge bosons that mediate strong interactions of quarks in quantum chromodynamics (QCD).
Diagram 1: In Feynman diagrams, emitted gluons are represented as helices. This diagram depicts the annihilation of an electron and positron.
Note that the blue wave is depicting Gamma ray photons and q ,q¯ are the quark, antiquark.
https://en.wikipedia.org/wiki/Antiparticle
Retrieved: 9 September 2017
Archive: https://archive.is/oKbuN
Antiparticle
In particle physics, corresponding to most kinds of particles there is an associated antiparticle with the same mass and opposite charge (including electric charge). For example, the antiparticle of the electron is the positron (antielectron), which has positive charge and is produced naturally in certain types of radioactive decay. The opposite is also true: the antiparticle of the positron is the electron.
Some particles, such as the photon, are their own antiparticle. Otherwise, for each pair of antiparticle partners, one is designated as normal matter (the kind we are made of), and the other (usually given the prefix "anti-") as in antimatter.
Particle–antiparticle pairs can annihilate each other, producing photons; since the charges of the particle and antiparticle are opposite, total charge is conserved. For example, the positrons produced in natural radioactive decay quickly annihilate themselves with electrons, producing pairs of gamma rays, a process exploited in positron emission tomography.
The laws of nature are very nearly symmetrical with respect to particles and antiparticles. For example, an antiproton and a positron can form an antihydrogen atom, which is believed to have the same properties as a hydrogen atom. This leads to the question of why the formation of matter after the Big Bang resulted in a universe consisting almost entirely of matter, rather than being a half-and-half mixture of matter and antimatter. The discovery of Charge Parity violation helped to shed light on this problem by showing that this symmetry, originally thought to be perfect, was only approximate.
Illustration of electric charge of particles (left) and antiparticles (right). From top to bottom; electron/positron, proton/antiproton, neutron/antineutron.
https://en.wikipedia.org/wiki/Antimatter
Retrieved: 9 September 2017
Archive: https://archive.is/8iNrR
Antimatter
In modern physics, antimatter is defined as a material composed of the antiparticle (or "partners") to the corresponding particles of ordinary matter.
A particle and its anti-particle have -in theory- the same mass as one another, but opposite electric charge, and other differences in quantum numbers. For example, a proton has positive charge while an antiproton has negative charge. A collision between any particle and its anti-particle partner is known to lead to their mutual annihilation, giving rise to various proportions of intense photons (gamma rays), neutrinos, and sometimes less-massive particle–antiparticle pairs.
Annihilation usually results in a release of energy that becomes available for heat or work. The amount of the released energy is usually proportional to the total mass of the collided matter and antimatter, in accord with the mass–energy equivalence equation, E = mc2.[1]
Antimatter particles bind with one another to form antimatter, just as ordinary particles bind to form normal matter. For example, a positron (the antiparticle of the electron) and an antiproton (the antiparticle of the proton) can form an antihydrogen atom. Physical principles indicate that complex antimatter atomic nuclei are possible, as well as anti-atoms corresponding to the known chemical elements.
There is considerable speculation as to why the observable universe is composed almost entirely of ordinary matter, as opposed to an equal mixture of matter and antimatter. This asymmetry of matter and antimatter in the visible universe is one of the great unsolved problems in physics. [2] The process by which this inequality between matter antimatter particles developed is called baryogenesis.
MES Note: How dare they equate the asymmetry problem with the truly great unsolved problem of 9/11! #FreeEnergy
Antimatter in the form of anti-atoms is one of the most difficult materials to produce. Individual antimatter particles, however, are commonly produced by particle accelerators and in some types of radioactive decay. The nuclei of antihelium have been artificially produced with difficulty. These are the most complex anti-nuclei so far observed.[3]
https://commons.wikimedia.org/w/index.php?title=File%3AAntimatter_Explosions_2.ogv
Retrieved: 9 September 2017
Archive: https://archive.is/DhUIj
Note: TGF = Terrestrial Gamma-ray Flash
…
The Feynman–Stueckelberg interpretation states that antimatter and antiparticles are regular particles traveling backward in time.[13]
Notation[edit]
One way to denote an antiparticle is by adding a bar over the particle's symbol. For example, the proton and antiproton are denoted as p and p¯, respectively. The same rule applies if one were to address a particle by its constituent components. A proton is made up of u?u?d quarks, so an antiproton must therefore be formed from uud antiquarks. Another convention is to distinguish particles by their electric charge. Thus, the electron and positron are denoted simply as e- and e+ respectively. However, to prevent confusion, the two conventions are never mixed.
https://en.wikipedia.org/wiki/Positron
Retrieved: 9 September 2017
Archive: https://archive.is/iXbLI
Positron
The positron or antielectron is the antiparticle or the antimatter counterpart of the electron. The positron has an electric charge of +1 e, a spin of 1/2 (same as electron), and has the same mass as an electron. When a positron collides with an electron, annihilation occurs. If this collision occurs at low energies, it results in the production of two or more gamma ray photons (see electron–positron annihilation).
Cloud chamber photograph of the first positron ever observed Original caption: A 63 million volt positron (H? = 2.1×105 gauss-cm) passing through a 6 mm lead plate and emerging as a 23 million volt positron (H? = 7.5×104 gauss-cm). The length of this latter path is at least ten times greater than the possible length of a proton path of this curvature
.
https://www.quora.com/What-will-happen-if-I-send-an-electron-back-in-time
Retrieved: 9 September 2017
Archive: https://archive.is/Uu0aG
https://en.wikipedia.org/wiki/Electric_charge
Retrieved: 9 September 2017
Archive: https://archive.is/h33cd
Electric charge
Electric charge is the physical property of matter that causes it to experience a force when placed in an electromagnetic field. There are two types of electric charges: positive and negative (commonly carried by protons and electrons respectively). Like charges repel and unlike attract. An absence of net charge is referred to as neutral. An object is negatively charged if it has an excess of electrons, and is otherwise positively charged or uncharged. The SI derived unit of electric charge is the coulomb (C). In electrical engineering, it is also common to use the ampere-hour (Ah), and, in chemistry, it is common to use the elementary charge (e) as a unit. The symbol Q often denotes charge. Early knowledge of how charged substances interact is now called classical electrodynamics, and is still accurate for problems that don't require consideration of quantum effects.
The electric charge is a fundamental conserved property of some subatomic particles, which determines their electromagnetic interaction. Electrically charged matter is influenced by, and produces, electromagnetic fields. The interaction between a moving charge and an electromagnetic field is the source of the electromagnetic force, which is one of the four fundamental forces (See also: magnetic field).
Twentieth-century experiments demonstrated that electric charge is quantized; that is, it comes in integer multiples of individual small units called the elementary charge, e, approximately equal to 1.602×10-19 coulombs (except for particles called quarks, which have charges that are integer multiples of 1/3e). The proton has a charge of +e, and the electron has a charge of -e. The study of charged particles, and how their interactions are mediated by photons, is called quantum electrodynamics.
Electric field of a positive and a negative point charge.
https://en.wikipedia.org/wiki/Electricity
Retrieved: 10 September 2017
Archive: https://archive.is/U0Vjx
Electricity
Electricity is the set of physical phenomena associated with the presence of electric charge. Although initially considered a phenomenon separate from magnetism, since the development of Maxwell's equations, both are recognized as part of a single phenomenon: electromagnetism. Various common phenomena are related to electricity, including lightning, static electricity, electric heating, electric discharges and many others. In addition, electricity is at the heart of many modern technologies.
The presence of an electric charge, which can be either positive or negative, produces an electric field. On the other hand, the movement of electric charges, which is known as electric current, produces a magnetic field.
When a charge is placed in a location with non-zero electric field, a force will act on it. The magnitude of this force is given by Coulomb's law. Thus, if that charge were to move, the electric field would be doing work on the electric charge. Thus we can speak of electric potential at a certain point in space, which is equal to the work done by an external agent in carrying a unit of positive charge from an arbitrarily chosen reference point to that point without any acceleration and is typically measured in volts.
In electrical engineering, electricity is used for:• electric power where electric current is used to energise equipment;
• electronics which deals with electrical circuits that involve active electrical components such as vacuum tubes, transistors, diodes and integrated circuits, and associated passive interconnection technologies.Electrical phenomena have been studied since antiquity, though progress in theoretical understanding remained slow until the seventeenth and eighteenth centuries. Even then, practical applications for electricity were few, and it would not be until the late nineteenth century that engineers were able to put it to industrial and residential use. The rapid expansion in electrical technology at this time transformed industry and society. Electricity's extraordinary versatility means it can be put to an almost limitless set of applications which include transport, heating, lighting, communications, and computation. Electrical power is now the backbone of modern industrial society.[1]
…
Magnetic field circles around a current
A moving charge (i.e. current) creates a magnetic field.
Moving charged elementary particles would thus also create a magnetic field, called a magnetic moment.
https://en.wikipedia.org/wiki/Torque
Retrieved: 14 September 2017
Archive: https://archive.is/wtaiQ
Torque
Torque, moment, or moment of force (see "Defining terminology" below) is rotational force.[1] Just as a linear force is a push or a pull, a torque can be thought of as a twist to an object.
https://en.wikipedia.org/wiki/Magnetic_moment#Elementary_particles
Retrieved: 14 September 2017
Archive: https://archive.is/siwf4
Magnetic moment
The magnetic moment of a magnet is a quantity that determines the torque it will experience in an external magnetic field. A loop of electric current, a bar magnet, an electron, a molecule, and a planet all have magnetic moments.
…
Elementary particles[edit]
In atomic and nuclear physics, the Greek symbol µ represents the magnitude of the magnetic moment, often measured in Bohr magnetons or nuclear magnetons, associated with the intrinsic spin of the particle and/or with the orbital motion of the particle in a system.
https://en.wikipedia.org/wiki/Magnetism
Retrieved: 10 September 2017
Archive: https://archive.is/abR8R
Magnetism
Magnetism is a class of physical phenomena that are mediated by magnetic fields. Electric currents and the magnetic moments of elementary particles give rise to a magnetic field, which acts on other currents and magnetic moments. The most familiar effects occur in ferromagnetic materials, which are strongly attracted by magnetic fields and can be magnetized to become permanent magnets, producing magnetic fields themselves. Only a few substances are ferromagnetic; the most common ones are iron, nickel and cobalt and their alloys. The prefix ferro- refers to iron, because permanent magnetism was first observed in lodestone, a form of natural iron ore called magnetite, Fe3O4.
…
History
Magnetism was first discovered in the ancient world, when people noticed that lodestones, naturally magnetized pieces of the mineral magnetite, could attract iron.[1] The word magnet comes from the Greek term µa???t?? ????? magnetis lithos,[2] "the Magnesian stone,[3] lodestone." In ancient Greece, Aristotle attributed the first of what could be called a scientific discussion of magnetism to the philosopher Thales of Miletus, who lived from about 625 BC to about 545 BC.[4] Around the same time, in ancient India, the Indian surgeon Sushruta was the first to make use of the magnet for surgical purposes.[5]
Lodestone, a natural magnet, attracting iron nails. Ancient humans discovered the property of magnetism from lodestone.
…
In 1600, William Gilbert published his De Magnete, Magneticisque Corporibus, et de Magno Magnete Tellure (On the Magnet and Magnetic Bodies, and on the Great Magnet the Earth). In this work he describes many of his experiments with his model earth called the terrella. From his experiments, he concluded that the Earth was itself magnetic and that this was the reason compasses pointed north (previously, some believed that it was the pole star (Polaris) or a large magnetic island on the north pole that attracted the compass).
…
Sources of magnetism
Magnetism, at its root, arises from two sources:
- Electric current (see Electron magnetic moment).
- Spin magnetic moments of elementary particles. The magnetic moments of the nuclei of atoms are typically thousands of times smaller than the electrons' magnetic moments, so they are negligible in the context of the magnetization of materials. Nuclear magnetic moments are nevertheless very important in other contexts, particularly in nuclear magnetic resonance (NMR) and magnetic resonance imaging (MRI).
…
Magnetic force
The phenomenon of magnetism is "mediated" by the magnetic field. An electric current or magnetic dipole creates a magnetic field, and that field, in turn, imparts magnetic forces on other particles that are in the fields.
Maxwell's equations, which simplify to the Biot–Savart law in the case of steady currents, describe the origin and behavior of the fields that govern these forces. Therefore, magnetism is seen whenever electrically charged particles are in motion—for example, from movement of electrons in an electric current, or in certain cases from the orbital motion of electrons around an atom's nucleus. They also arise from "intrinsic" magnetic dipoles arising from quantum-mechanical spin.
The same situations that create magnetic fields—charge moving in a current or in an atom, and intrinsic magnetic dipoles—are also the situations in which a magnetic field has an effect, creating a force.
Magnetic lines of force of a bar magnet shown by iron filings on paper
…
Magnetic dipoles
A very common source of magnetic field found in nature is a dipole, with a "South pole" and a "North pole", terms dating back to the use of magnets as compasses, interacting with the Earth's magnetic field to indicate North and South on the globe. Since opposite ends of magnets are attracted, the north pole of a magnet is attracted to the south pole of another magnet. The Earth's North Magnetic Pole (currently in the Arctic Ocean, north of Canada) is physically a south pole, as it attracts the north pole of a compass. A magnetic field contains energy, and physical systems move toward configurations with lower energy.
Wait, the North Pole is actually a south pole??!!
https://en.wikipedia.org/wiki/Earth%27s_magnetic_field
Retrieved: 10 September 2017
Archive: https://archive.is/nIqOp
Earth's magnetic field
Earth's magnetic field, also known as the geomagnetic field, is the magnetic field that extends from the Earth's interior out into space, where it meets the solar wind, a stream of charged particles emanating from the Sun. Its magnitude at the Earth's surface ranges from 25 to 65 microteslas (0.25 to 0.65 gauss).[3]Roughly speaking it is the field of a magnetic dipole currently tilted at an angle of about 11 degrees with respect to Earth's rotational axis, as if there were a bar magnet placed at that angle at the center of the Earth. The North geomagnetic pole, located near Greenland in the northern hemisphere, is actually the south pole of the Earth's magnetic field, and the South geomagnetic pole is the north pole. Unlike a bar magnet, Earth's magnetic field changes over time because it is generated by a geodynamo (in Earth's case, the motion of molten iron alloys in its outer core).
While the North and South magnetic poles are usually located near the geographic poles, they can wander widely over geological time scales, but sufficiently slowly for ordinary compasses to remain useful for navigation. However, at irregular intervals averaging several hundred thousand years, the Earth's field reverses and the North and South Magnetic Poles relatively abruptly switch places. These reversals of the geomagnetic poles leave a record in rocks that are of value to paleomagnetists in calculating geomagnetic fields in the past. Such information in turn is helpful in studying the motions of continents and ocean floors in the process of plate tectonics.
The magnetosphere is the region above the ionosphere that is defined by the extent of the Earth's magnetic field in space. It extends several tens of thousands of kilometers into space, protecting the Earth from the charged particles of the solar wind and cosmic rays that would otherwise strip away the upper atmosphere, including the ozone layer that protects the Earth from harmful ultraviolet radiation.
Computer simulation of the Earth's field in a period of normal polarity between reversals.[1] The lines represent magnetic field lines, blue when the field points towards the center and yellow when away. The rotation axis of the Earth is centered and vertical. The dense clusters of lines are within the Earth's core.[2]
…
…
An artist's rendering of the structure of a magnetosphere. 1) Bow shock. 2) Magnetosheath. 3) Magnetopause. 4) Magnetosphere. 5) Northern tail lobe. 6) Southern tail lobe. 7) Plasmasphere.
…
A schematic illustrating the relationship between motion of conducting fluid, organized into rolls by the Coriolis force, and the magnetic field the motion generates.[42]
https://en.wikipedia.org/wiki/Electromagnetic_field
Retrieved: 12 September 2017
Archive: https://archive.is/BP7qM
Electromagnetic field
An electromagnetic field (also EMF or EM field) is a physical field produced by electrically charged objects.[1] It affects the behavior of charged objects in the vicinity of the field. The electromagnetic field extends indefinitely throughout space and describes the electromagnetic interaction. It is one of the four fundamental forces of nature (the others are gravitation, weak interaction and strong interaction).
The field can be viewed as the combination of an electric field and a magnetic field. The electric field is produced by stationary charges, and the magnetic field by moving charges (currents); these two are often described as the sources of the field. The way in which charges and currents interact with the electromagnetic field is described by Maxwell's equations and the Lorentz force law.
From a classical perspective in the history of electromagnetism, the electromagnetic field can be regarded as a smooth, continuous field, propagated in a wavelike manner; whereas from the perspective of quantum field theory, the field is seen as quantized, being composed of individual particles.
Structure[edit]
The electromagnetic field may be viewed in two distinct ways: a continuous structure or a discrete structure.
Continuous structure[edit]
Classically, electric and magnetic fields are thought of as being produced by smooth motions of charged objects. For example, oscillating charges produce electric and magnetic fields that may be viewed in a 'smooth', continuous, wavelike fashion. In this case, energy is viewed as being transferred continuously through the electromagnetic field between any two locations. For instance, the metal atoms in a radio transmitter appear to transfer energy continuously. This view is useful to a certain extent (radiation of low frequency), but problems are found at high frequencies (see ultraviolet catastrophe).[citation needed]
Discrete structure[edit]
The electromagnetic field may be thought of in a more 'coarse' way. Experiments reveal that in some circumstances electromagnetic energy transfer is better described as being carried in the form of packets called quanta (in this case, photons) with a fixed frequency.
https://en.wikipedia.org/wiki/Electric_field
Retrieved: 13 September 2017
Archive: https://archive.is/ZFwbk
Electric field
An electric field is a vector field that associates to each point in space the Coulomb force that would be experienced per unit of electric charge, by an infinitesimal test charge at that point.[1] Electric fields are created by electric charges and can be induced by time-varying magnetic fields. The electric field combines with the magnetic field to form the electromagnetic field.
Electric field lines emanating from a point positive electric charge suspended over an infinite sheet of conducting material.
https://en.wikipedia.org/wiki/Coulomb%27s_law
Retrieved: 13 September 2017
Archive: https://archive.is/cCFbD
Coulomb's law
Coulomb's law, or Coulomb's inverse-square law, is a law of physics that describes force interacting between static electrically charged particles. In its scalar form, the law is:
where ke is Coulomb's constant (ke = 8.99×109 N m2 C-2), q1 and q2 are the signed magnitudes of the charges, and the scalar r is the distance between the charges. The force of interaction between the charges is attractive if the charges have opposite signs (i.e., F is negative) and repulsive if like-signed (i.e., F is positive).
The law was first published in 1784 by French physicist Charles-Augustin de Coulomb and was essential to the development of the theory of electromagnetism. Being an inverse-square law, it is analogous to Isaac Newton's inverse-square law of universal gravitation. Coulomb's law can be used to derive Gauss's law, and vice versa. The law has been tested extensively, and all observations have upheld the law's principle.
…
Experiment to verify Coulomb's law.
https://en.wikipedia.org/wiki/Magnetic_field
Retrieved: 13 September 2017
Archive: https://archive.is/am5Tw
Magnetic field
A magnetic field is the magnetic effect of electric currents and magnetic materials. The magnetic field at any given point is specified by both a direction and a magnitude (or strength); as such it is represented by a vector field.[nb 1] The term is used for two distinct but closely related fields denoted by the symbols B and H, where H is measured in units of amperes per meter (symbol: A·m-1 or A/m) in the SI. B is measured in teslas (symbol: T) and newtons per meter per ampere (symbol: N·m-1·A-1 or N/(m·A)) in the SI. B is most commonly defined in terms of the Lorentz force it exerts on moving electric charges.
Magnetic fields can be produced by moving electric charges and the intrinsic magnetic moments of elementary particles associated with a fundamental quantum property, their spin.[1][2] In special relativity, electric and magnetic fields are two interrelated aspects of a single object, called the electromagnetic tensor; the split of this tensor into electric and magnetic fields depends on the relative velocity of the observer and charge. In quantum physics, the electromagnetic field is quantized and electromagnetic interactions result from the exchange of photons.
In everyday life, magnetic fields are most often encountered as a force created by permanent magnets, which pull on ferromagnetic materials such as iron, cobalt, or nickel, and attract or repel other magnets.
Magnetic fields are widely used throughout modern technology, particularly in electrical engineering and electromechanics. The Earth produces its own magnetic field, which is important in navigation, and it shields the Earth's atmosphere from solar wind. Rotating magnetic fields are used in both electric motors and generators. Magnetic forces give information about the charge carriers in a material through the Hall effect. The interaction of magnetic fields in electric devices such as transformers is studied in the discipline of magnetic circuits.
Magnetic field of an ideal cylindrical magnet with its axis of symmetry inside the image plane. The magnetic field is represented by magnetic field lines, which show the direction of the field at different points.
…
Compasses reveal the direction of the local magnetic field. As seen here, the magnetic field points towards a magnet's south pole and away from its north pole.
…
Magnetic field and electric currents
Currents of electric charges both generate a magnetic field and feel a force due to magnetic B-fields.
…
Right hand grip rule: a current flowing in the direction of the white arrow produces a magnetic field shown by the red arrows.
Note: I believe the current moving from North to South is simply by convention, and could theoretically have been the other way around.
http://hyperphysics.phy-astr.gsu.edu/hbase/magnetic/magfield.html
Retrieved: 13 September 2017
Archive: https://archive.is/DeqzM
Magnetic Field Strength H
The magnetic fields generated by currents and calculated from Ampere's Law or the Biot-Savart Law are characterized by the magnetic field B measured in Tesla. But when the generated fields pass through magnetic materials which themselves contribute internal magnetic fields, ambiguities can arise about what part of the field comes from the external currents and what comes from the material itself. It has been common practice to define another magnetic field quantity, usually called the "magnetic field strength" designated by H.
https://en.wikipedia.org/wiki/Electric_current
Retrieved: 13 September 2017
Archive: https://archive.is/xhSFG
Electric current
An electric current is a flow of electric charge.[1]:2 In electric circuits this charge is often carried by moving electrons in a wire. It can also be carried by ions in an electrolyte, or by both ions and electrons such as in an ionised gas (plasma).[2]
The SI unit for measuring an electric current is the ampere, which is the flow of electric charge across a surface at the rate of one coulomb per second. Electric current is measured using a device called an ammeter.[3]
Electric currents cause Joule heating, which creates light in incandescent light bulbs. They also create magnetic fields, which are used in motors, inductors and generators.The moving charged particles in an electric current are called charge carriers. In metals, one or more electrons from each atom are loosely bound to the atom, and can move freely about within the metal. These conduction electrons are the charge carriers in metal conductors.
A simple electric circuit, where current is represented by the letter i. The relationship between the voltage (V), resistance (R), and current (I) is V=IR; this is known as Ohm's law.
…
Conventions
In a conductive material, the moving charged particles which constitute the electric current are called charge carriers. In metals, which make up the wires and other conductors in most electrical circuits, the positively charged atomic nuclei are held in a fixed position, and the negatively charged electrons are free to move, carrying their charge from one place to another. In other materials, notably the semiconductors, the charge carriers can be positive or negative, depending on the dopant used. Positive and negative charge carriers may even be present at the same time, as happens in an electrolyte in an electrochemical cell.
A flow of positive charges gives the same electric current, and has the same effect in a circuit, as an equal flow of negative charges in the opposite direction. Since current can be the flow of either positive or negative charges, or both, a convention is needed for the direction of current that is independent of the type of charge carriers. The direction of conventional current is arbitrarily defined as the same direction as positive charges flow.
…
The electrons, the charge carriers in an electrical circuit, flow in the opposite direction of the conventional electric current.
https://en.wikipedia.org/wiki/Electromagnetic_radiation
Retrieved: 11 September 2017
Archive: https://archive.is/cbf8T
Electromagnetic radiation
In physics, electromagnetic radiation (EM radiation or EMR) refers to the waves (or their quanta, photons) of the electromagnetic field, propagating (radiating) through space carrying electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visible) light, ultraviolet, X-, and gamma ray.
Classically, electromagnetic radiation consists of electromagnetic waves, which are synchronized oscillations of electric and magnetic fields that propagate at the speed of light through a vacuum. The oscillations of the two fields are perpendicular to each other and perpendicular to the direction of energy and wave propagation, forming a transverse wave. The wavefront of electromagnetic waves emitted from a point source (such as a lightbulb) is a sphere. The position of an electromagnetic wave within the electromagnetic spectrum could be characterized by either its frequency of oscillation or its wavelength. The electromagnetic spectrum includes, in order of increasing frequency and decreasing wavelength: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.[1]
Electromagnetic waves are produced whenever charged particles are accelerated, and these waves can subsequently interact with other charged particles. EM waves carry energy, momentum and angular momentum away from their source particle and can impart those quantities to matter with which they interact. Quanta of EM waves are called photons, whose rest mass is zero, but whose energy, or equivalent total (relativistic) mass, is not zero so they are still affected by gravity. Electromagnetic radiation is associated with those EM waves that are free to propagate themselves ("radiate") without the continuing influence of the moving charges that produced them, because they have achieved sufficient distance from those charges. Thus, EMR is sometimes referred to as the far field. In this language, the near field refers to EM fields near the charges and current that directly produced them, specifically, electromagnetic induction and electrostatic induction phenomena.
In the quantum theory of electromagnetism[2], EMR consists of photons, the elementary particles responsible for all electromagnetic interactions[3]. Quantum effects provide additional sources of EMR, such as the transition of electrons to lower energy levels in an atom and black-body radiation. The energy of an individual photon is quantized and is greater for photons of higher frequency.
…
The electromagnetic waves that compose electromagnetic radiation can be imagined as a self-propagating transverse oscillating wave of electric and magnetic fields. This diagram shows a plane linearly polarized EMR wave propagating from left to right (X axis). The electric field is in a vertical plane (Z axis) and the magnetic field in a horizontal plane (Y axis). The electric and magnetic fields in EMR waves are always in phase and at 90 degrees to each other.
…
Electromagnetic spectrum with visible light highlighted
…
Ultraviolet[edit]
As frequency increases into the ultraviolet, photons now carry enough energy (about three electron volts or more) to excite certain doubly bonded molecules into permanent chemical rearrangement. In DNA, this causes lasting damage. DNA is also indirectly damaged by reactive oxygen species produced by ultraviolet A (UVA), which has energy too low to damage DNA directly. This is why ultraviolet at all wavelengths can damage DNA, and is capable of causing cancer, and (for UVB) skin burns (sunburn) that are far worse than would be produced by simple heating (temperature increase) effects. This property of causing molecular damage that is out of proportion to heating effects, is characteristic of all EMR with frequencies at the visible light range and above. These properties of high-frequency EMR are due to quantum effects that permanently damage materials and tissues at the molecular level.[citation needed]
At the higher end of the ultraviolet range, the energy of photons becomes large enough to impart enough energy to electrons to cause them to be liberated from the atom, in a process called photoionisation. The energy required for this is always larger than about 10 electron volts (eV) corresponding with wavelengths smaller than 124 nm (some sources suggest a more realistic cutoff of 33 eV, which is the energy required to ionize water). This high end of the ultraviolet spectrum with energies in the approximate ionization range, is sometimes called "extreme UV." Ionizing UV is strongly filtered by the Earth's atmosphere).[citation needed]
…
Atmosphere and ionosphere
Most UV and X-rays are blocked by absorption first from molecular nitrogen, and then (for wavelengths in the upper UV) from the electronic excitation of dioxygen and finally ozone at the mid-range of UV. Only 30% of the Sun's ultraviolet light reaches the ground, and almost all of this is well transmitted.
Visible light is well transmitted in air, as it is not energetic enough to excite nitrogen, oxygen, or ozone, but too energetic to excite molecular vibrational frequencies of water vapor.[citation needed]
Absorption bands in the infrared are due to modes of vibrational excitation in water vapor. However, at energies too low to excite water vapor, the atmosphere becomes transparent again, allowing free transmission of most microwave and radio waves.[citation needed]
Finally, at radio wavelengths longer than 10 meters or so (about 30 MHz), the air in the lower atmosphere remains transparent to radio, but plasma in certain layers of the ionosphere begins to interact with radio waves (see skywave). This property allows some longer wavelengths (100 meters or 3 MHz) to be reflected and results in shortwave radio beyond line-of-sight. However, certain ionospheric effects begin to block incoming radiowaves from space, when their frequency is less than about 10 MHz (wavelength longer than about 30 meters).[citation needed]
Rough plot of Earth's atmospheric absorption and scattering (or opacity) of various wavelengths of electromagnetic radiation
https://en.wikipedia.org/wiki/Light
Retrieved: 11 September 2017
Archive: https://archive.is/NU5hG
Light
Light is electromagnetic radiation within a certain portion of the electromagnetic spectrum. The word usually refers to visible light, which is visible to the human eye and is responsible for the sense of sight.[1] Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), or 4.00 × 10-7 to 7.00 × 10-7 m, between the infrared (with longer wavelengths) and the ultraviolet (with shorter wavelengths).[2][3] This wavelength means a frequency range of roughly 430–750 terahertz (THz).
The main source of light on Earth is the Sun. Sunlight provides the energy that green plants use to create sugars mostly in the form of starches, which release energy into the living things that digest them. This process of photosynthesis provides virtually all the energy used by living things. Historically, another important source of light for humans has been fire, from ancient campfires to modern kerosene lamps. With the development of electric lights and power systems, electric lighting has effectively replaced firelight. Some species of animals generate their own light, a process called bioluminescence. For example, fireflies use light to locate mates, and vampire squids use it to hide themselves from prey.
The primary properties of visible light are intensity, propagation direction, frequency or wavelength spectrum, and polarization, while its speed in a vacuum, 299,792,458 metres per second, is one of the fundamental constants of nature. Visible light, as with all types of electromagnetic radiation (EMR), is experimentally found to always move at this speed in a vacuum.[citation needed]
In physics, the term light sometimes refers to electromagnetic radiation of any wavelength, whether visible or not.[4][5] In this sense, gamma rays, X-rays, microwaves and radio waves are also light. Like all types of light, visible light is emitted and absorbed in tiny "packets" called photons and exhibits properties of both waves and particles. This property is referred to as the wave–particle duality. The study of light, known as optics, is an important research area in modern physics.
A triangular prism dispersing a beam of white light. The longer wavelengths (red) and the shorter wavelengths (blue) are separated.
Electromagnetic spectrum and visible light
Generally, EM radiation, or EMR (the designation "radiation" excludes static electric and magnetic and near fields), is classified by wavelength into radio, microwave, infrared, the visible region that we perceive as light, ultraviolet, X-rays and gamma rays.
The behavior of EMR depends on its wavelength. Higher frequencies have shorter wavelengths, and lower frequencies have longer wavelengths. When EMR interacts with single atoms and molecules, its behavior depends on the amount of energy per quantum it carries.
EMR in the visible light region consists of quanta (called photons) that are at the lower end of the energies that are capable of causing electronic excitation within molecules, which leads to changes in the bonding or chemistry of the molecule. At the lower end of the visible light spectrum, EMR becomes invisible to humans (infrared) because its photons no longer have enough individual energy to cause a lasting molecular change (a change in conformation) in the visual molecule retinal in the human retina, which change triggers the sensation of vision.
Electromagnetic spectrum with light highlighted
…
Speed of light
The speed of light in a vacuum is defined to be exactly 299,792,458 m/s (approx. 186,282 miles per second). The fixed value of the speed of light in SI units results from the fact that the metre is now defined in terms of the speed of light. All forms of electromagnetic radiation move at exactly this same speed in vacuum.
Recall from Part 1, in Rupert Sheldrake's censored TED Talk he talked about the possibility of the speed of light NOT being constant and how mainstream science is dogmatic in their definitions.
My current speculation is that the speed of light not being constant may have something to do with the hidden science involved in hidden #FreeEnergy technology…
https://en.wikipedia.org/wiki/Near_and_far_field
Retrieved: 16 September 2017
Archive: https://archive.is/3rwce
Near and far field
The near field and far field are regions of the electromagnetic field around an object, such as a transmitting antenna, or the result of radiation scattering off an object. Non-radiative 'near-field' behaviours of electromagnetic fields dominate close to the antenna or scattering object, while electromagnetic radiation 'far-field' behaviours dominate at greater distances.
Differences between Fraunhofer diffraction and Fresnel diffraction
https://en.wikipedia.org/wiki/Dispersive_prism
Retrieved: 11 September 2017
Archive: https://archive.is/5W3xP
Dispersive prism
In optics, a dispersive prism is a type of optical prism, usually having the shape of a geometrical triangular prism. It is the most widely known type of optical prism, although perhaps not the most common in actual use. Triangular prisms are used to disperse light, that is, to break light up into its spectral components (the colors of the rainbow). This dispersion occurs because the angle of refraction is dependent on the refractive index of a certain material which in turn is slightly dependent on the wavelength of light that is travelling through it. This means that different wavelengths of light will travel at different speeds, and so the light will disperse into the colours of the visible spectrum, with longer wavelengths (red, yellow) being refracted less than shorter wavelengths (violet, blue).
Photograph of a triangular prism, dispersing light
https://en.wikipedia.org/wiki/Spectrum
Retrieved: 11 September 2017
Archive: https://archive.is/IYJPt
Spectrum
A spectrum (plural spectra or spectrums[1]) is a condition that is not limited to a specific set of values but can vary, without steps, across a continuum. The word was first used scientifically in optics to describe the rainbow of colors in visible light after passing through a prism. As scientific understanding of light advanced, it came to apply to the entire electromagnetic spectrum.
…
Electromagnetic spectrum
…
Light from many different sources contains various colors, each with its own brightness or intensity. A rainbow, or prism, sends these component colors in different directions, making them individually visible at different angles. A graph of the intensity plotted against the frequency (showing the brightness of each color) is the frequency spectrum of the light. When all the visible frequencies are present equally, the perceived color of the light is white, and the spectrum is a flat line. Therefore, flat-line spectrums in general are often referred to as white, whether they represent light or another type of wave phenomenon (sound, for example, or vibration in a structure).
In radio and telecommunications, the frequency spectrum can be shared among many different broadcasters. The radio spectrum is the part of the electromagnetic spectrum corresponding to frequencies lower below 300 GHz, which corresponds to wavelengths longer than about 1 mm. The microwave spectrum corresponds to frequencies between 300 MHz (0.3 GHz) and 300 GHz and wavelengths between one meter and one millimeter.[5][6] Each broadcast radio and TV station transmits a wave on an assigned frequency range, called a channel. When many broadcasters are present, the radio spectrum consists of the sum of all the individual channels, each carrying separate information, spread across a wide frequency spectrum. Any particular radio receiver will detect a single function of amplitude (voltage) vs. time. The radio then uses a tuned circuit or tuner to select a single channel or frequency band and demodulate or decode the information from that broadcaster. If we made a graph of the strength of each channel vs. the frequency of the tuner, it would be the frequency spectrum of the antenna signal.
In astronomy, stellar classification is the classification of stars based on their characteristic electromagnetic spectra. The spectral flux density is used to represent the spectrum of a light-source, such as a star.
In radiometry and colorimetry (or color science more generally), the spectral power distribution (SPD) of a light source is a measure of the power contributed by each frequency or color in a light source.
https://en.wikipedia.org/wiki/Radiant_energy
Retrieved: 11 September 2017
Archive: https://archive.is/CPt5Q
Radiant energy
In physics, and in particular as measured by radiometry, radiant energy is the energy of electromagnetic and gravitational radiation.[1] As energy, its SI unit is the joule (J). The quantity of radiant energy may be calculated by integrating radiant flux (or power) with respect to time. The symbol Qe is often used throughout literature to denote radiant energy ("e" for "energetic", to avoid confusion with photometric quantities). In branches of physics other than radiometry, electromagnetic energy is referred to using E or W. The term is used particularly when electromagnetic radiation is emitted by a source into the surrounding environment. This radiation may be visible or invisible to the human eye.[2][3]
Visible light such as sunlight carries radiant energy, which is used in solar power generation.
…
Analysis
Because electromagnetic (EM) radiation can be conceptualized as a stream of photons, radiant energy can be viewed as photon energy – the energy carried by these photons. Alternatively, EM radiation can be viewed as an electromagnetic wave, which carries energy in its oscillating electric and magnetic fields. These two views are completely equivalent and are reconciled to one another in quantum field theory (see wave-particle duality).
https://en.wikipedia.org/wiki/Photon
Retrieved: 9 September 2017
Archive: https://archive.is/NYrMW
Photon
A photon is a type of elementary particle, the quantum of the electromagnetic field including electromagnetic radiation such as light, and the force carrier for the electromagnetic force (even when static via virtual photons). The photon has zero rest mass and always moves at the speed of light within a vacuum.
Like all elementary particles, photons are currently best explained by quantum mechanics and exhibit wave–particle duality, exhibiting properties of both waves and particles. For example, a single photon may be refracted by a lens and exhibit wave interference with itself, and it can behave as a particle with definite and finite measurable position or momentum, though not both at the same time.
https://en.wikipedia.org/wiki/Photon_energy
Retrieved: 11 September 2017
Archive: https://archive.is/uVoSh
Photon energy
Photon energy is the energy carried by a single photon. The amount of energy is directly related to the photon's electromagnetic wavelength and frequency. The higher the photon's frequency, the higher its energy. Equivalently, the longer the photon's wavelength, the lower its energy.
Photon energy is solely a function of the photon's wavelength. Other factors, such as the intensity of the radiation, do not affect photon energy. In other words, two photons of light with the same color and therefore, same wavelength, will have the same photon energy, even if one was emitted from a wax candle and the other from the Sun.
Photon energy can be represented by any unit of energy. Among the units commonly used to denote photon energy are the electronvolt (eV) and the joule (as well as its multiples, such as the microjoule). As one joule equals 6.24 × 1018 eV, the larger units may be more useful in denoting the energy of photons with higher frequency and higher energy, such as gamma rays, as opposed to lower energy photons, such as those in the radiofrequency region of the electromagnetic spectrum.
If photons are in fact massless, photon energy would not related to mass through equivalence E = mc2. The only two observed kinds of so-called massless energetic particles are photons and gluons. [Citation Needed.] However, the claim that photons are massless is based on the crisis arising with other theories in quantum mechanics. In order for other math, such as gauge invariance and so-called "renormalization" to survive without considerable revision, photons must be kept massless in the realm of current equations. [1] The claim is also contested by other circles. [2] Photons are said to have relativistic mass (i.e. mass deriving from a material body's motion against another). Moreover, some hypotheses propose that all mass or "rest mass" might itself actually be composed of stacked relativistic mass, secondary to motion, since no material body is nor can be truly at "rest" relative to all fields. In this hypotheses, as motion becomes zero, mass also becomes zero. On the other hand, photons have motion and varying energy depending on the frequency and wavelength, suggesting that various forms of the photon each have different mass equivalence. Thus, "E = mc2" would show that mass and motion are inextricably linked and fundamentally interchangable concepts for all matter. [3]
Hmmmm VERY interesting information here regarding photons being massless yet having energy, and the mental gymnastics being played here to tie it in with Einstein's Mass-Energy equivalence formula…. # VERYInteresting!
https://en.wikipedia.org/wiki/Gamma_ray
Retrieved: 9 September 2017
Archive: https://archive.is/uGnQ2
Gamma ray
Gamma rays (also called gamma radiation), denoted by the lower-case Greek letter gamma (? or ?), are penetrating electromagnetic radiation of a kind arising from the radioactive decay of atomic nuclei. It consists of photons in the highest observed range of photon energy. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radium. In 1903, Ernest Rutherford named this radiation gamma rays. Rutherford had previously discovered two other types of radioactive decay, which he named alpha and beta rays.
Gamma rays are able to ionize atoms (ionizing radiation), and are thus biologically hazardous. The decay of an atomic nucleus from a high energy state to a lower energy state, a process called gamma decay, produces gamma radiation.
Natural sources of gamma rays on Earth are observed in the gamma decay of radionuclides and secondary radiation from atmospheric interactions with cosmic ray particles. There are rare terrestrial natural sources, such as lightning strikes and terrestrial gamma-ray flashes, that produce gamma rays not of a nuclear origin. Additionally, gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced, that in turn cause secondary gamma rays via bremsstrahlung, inverse Compton scattering, and synchrotron radiation. However, a large fraction of such astronomical gamma rays are screened by Earth's atmosphere and can only be detected by spacecraft. Gamma rays are produced by nuclear fusion in stars including the Sun (such as the CNO cycle), but are absorbed or inelastically scattered by the stellar material, reducing their energy, before escaping and are not observable from Earth as gamma rays.
Illustration of an emission of a gamma ray (?) from an atomic nucleus
Gamma rays are emitted during nuclear fission in nuclear explosions.
…
Radioactive decay (gamma decay)[edit]
Main article: Nuclear isomerGamma rays are produced during gamma decay, which normally occurs after other forms of decay occur, such as alpha or beta decay. An excited nucleus can decay by the emission of an a or ß particle. The daughter nucleus that results is usually left in an excited state. It can then decay to a lower energy state by emitting a gamma ray photon, in a process called gamma decay.
The emission of a gamma ray from an excited nucleus typically requires only 10-12 seconds. Gamma decay may also follow nuclear reactions such as neutron capture, nuclear fission, or nuclear fusion. Gamma decay is also a mode of relaxation of many excited states of atomic nuclei following other types of radioactive decay, such as beta decay, so long as these states possess the necessary component of nuclear spin.
…
Uses
…
In the US, gamma ray detectors are beginning to be used as part of the Container Security Initiative (CSI). These machines are advertised to be able to scan 30 containers per hour.
Gamma radiation is often used to kill living organisms, in a process called irradiation. Applications of this include the sterilization of medical equipment (as an alternative to autoclaves or chemical means), the removal of decay-causing bacteria from many foods and the prevention of the sprouting of fruit and vegetables to maintain freshness and flavor.
Despite their cancer-causing properties, gamma rays are also used to treat some types of cancer, since the rays also kill cancer cells.
Gamma-ray image of a truck with two stowaways taken with a VACIS (vehicle and container imaging system)
https://en.wikipedia.org/wiki/Ionizing_radiation
Retrieved: 14 September 2017
Archive: https://archive.is/tdmO1
Ionizing radiation
Ionizing radiation (ionising radiation) is radiation that carries enough energy to liberate electrons from atoms or molecules, thereby ionizing them. Ionizing radiation is made up of energetic subatomic particles, ions or atoms moving at high speeds (usually greater than 1% of the speed of light), and electromagnetic waves on the high-energy end of the electromagnetic spectrum.
Gamma rays, X-rays, and the higher ultraviolet part of the electromagnetic spectrum are ionizing, whereas the lower ultraviolet part of the electromagnetic spectrum, and also the lower part of the spectrum below UV, including visible light (including nearly all types of laser light), infrared, microwaves, and radio waves are all considered non-ionizing radiation. The boundary between ionizing and non-ionizing electromagnetic radiation that occurs in the ultraviolet is not sharply defined, since different molecules and atoms ionize at different energies. Conventional definition places the boundary at a photon energy between 10 eV and 33 eV in the ultraviolet (see definition boundary section below).
Typical ionizing subatomic particles from radioactivity include alpha particles, beta particles and neutrons. Almost all products of radioactive decay are ionizing because the energy of radioactive decay is typically far higher than that required to ionize. Other subatomic ionizing particles which occur naturally are muons, mesons, positrons, and other particles that constitute the secondary cosmic rays that are produced after primary cosmic rays interact with Earth's atmosphere.[1][2] Cosmic rays are generated by stars and certain celestial events such as supernova explosions. Cosmic rays may also produce radioisotopes on Earth (for example, carbon-14), which in turn decay and produce ionizing radiation. Cosmic rays and the decay of radioactive isotopes are the primary sources of natural ionizing radiation on Earth referred to as background radiation. Ionizing radiation can also be generated artificially using X-ray tubes, particle accelerators, and any of the various methods that produce radioisotopes artificially.
Ionizing radiation is not detectable by human senses, so radiation detection instruments such as Geiger counters must be used to indicate its presence and measure it. However, high intensities can cause emission of visible light upon interaction with matter, such as in Cherenkov radiation and radioluminescence. Ionizing radiation is used in a wide variety of fields such as medicine, nuclear power, research, manufacturing, construction, and many other areas, but presents a health hazard if proper measures against undesired exposure aren't followed. Exposure to ionizing radiation causes damage to living tissue, and can result in mutation, radiation sickness, cancer, and death.
Ionizing radiation hazard symbol
…
Alpha (a) radiation consists of a fast-moving helium-4 (4He) nucleus and is stopped by a sheet of paper. Beta (ß) radiation, consisting of electrons, is halted by an aluminium plate. Gamma (?) radiation, consisting of energetic photons, is eventually absorbed as it penetrates a dense material. Neutron (n) radiation consists of free neutrons that are blocked by light elements, like hydrogen, which slow and/or capture them. Not shown: galactic cosmic rays that consist of energetic charged nuclei such as protons, helium nuclei, and high-charged nuclei called HZE ions.
…
Different types of electromagnetic radiation
…
Radiation interaction: gamma rays are represented by wavy lines, charged particles and neutrons by straight lines. The small circles show where ionization occurs.
…
2007 ISO radioactivity danger symbol intended for IAEA Category 1, 2 and 3 sources defined as dangerous sources capable of death or serious injury.[35]
https://en.wikipedia.org/wiki/Radioactive_decay
Retrieved: 14 September 2017
Archive: https://archive.is/ytRgP
Radioactive decay
Radioactive decay (also known as nuclear decay or radioactivity) is the process by which an unstable atomic nucleus loses energy (in terms of mass in its rest frame) by emitting radiation, such as an alpha particle, beta particle with neutrino or only a neutrino in the case of electron capture, gamma ray, or electron in the case of internal conversion. A material containing such unstable nuclei is considered radioactive. Certain highly excited short-lived nuclear states can decay through neutron emission, or more rarely, proton emission.
Radioactive decay is a stochastic (i.e. random) process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay,[1][2][3]regardless of how long the atom has existed. However, for a collection of atoms, the collection's expected decay rate is characterized in terms of their measured decay constants or half-lives. This is the basis of radiometric dating. The half-lives of radioactive atoms have no known upper limit, spanning a time range of over 55 orders of magnitude, from nearly instantaneous to far longer than the age of the universe.
…
The decaying nucleus is called the parent radionuclide (or parent radioisotope[note 2]), and the process produces at least one daughter nuclide. Except for gamma decay or internal conversion from a nuclear excited state, the decay is a nuclear transmutation resulting in a daughter containing a different number of protons or neutrons (or both). When the number of protons changes, an atom of a different chemical element is created.
Taking an X-ray image with early Crookes tube apparatus in 1896. The Crookes tube is visible in the centre. The standing man is viewing his hand with a fluoroscope screen; this was a common way of setting up the tube. No precautions against radiation exposure are being taken; its hazards were not known at the time.
…
Radioactivity is characteristic of elements with large atomic number. Elements with at least one stable isotope are shown in light blue. Green shows elements whose most stable isotope has a half-life measured in millions of years. Yellow and orange are progressively less stable, with half-lives in thousands or hundreds of years, down toward one day. Red and purple show highly and extremely radioactive elements where the most stable isotopes exhibit half-lives measured on the order of one day and much less.
…
Alpha particles may be completely stopped by a sheet of paper, beta particles by aluminium shielding. Gamma rays can only be reduced by much more substantial mass, such as a very thick layer of lead.
https://en.wikipedia.org/wiki/X-ray
Retrieved: 14 September 2017
Archive: https://archive.is/r3DVW
X-ray
X-rays are part of the electromagnetic spectrum, with wavelengths shorter than visible light. Different applications use different parts of the X-ray spectrum.
https://en.wikipedia.org/wiki/Alpha_decay
Retrieved: 13 September 2017
Archive: https://archive.is/wAph9
Alpha decay
Alpha decay or a-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or 'decays' into an atom with a mass number that is reduced by four and an atomic number that is reduced by two. An alpha particle is identical to the nucleus of a helium-4 atom, which consists of two protons and two neutrons. It has a charge of +2e and a mass of 4u. For example, uranium-238 decays to form thorium-234. Alpha particles have a charge +2, but as a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms – the charge is not usually shown.
Alpha decay typically occurs in the heaviest nuclides. Theoretically, it can occur only in nuclei somewhat heavier than nickel (element 28), where the overall binding energy per nucleon is no longer a minimum and the nuclides are therefore unstable toward spontaneous fission-type processes. In practice, this mode of decay has only been observed in nuclides considerably heavier than nickel, with the lightest known alpha emitters being the lightest isotopes (mass numbers 106–110) of tellurium (element 52). Exceptionally, however, beryllium-8 decays to two alpha particles.
Alpha decay is by far the most common form of cluster decay, where the parent atom ejects a defined daughter collection of nucleons, leaving another defined product behind. It is the most common form because of the combined extremely high binding energy and relatively small mass of the alpha particle. Like other cluster decays, alpha decay is fundamentally a quantum tunneling process. Unlike beta decay, it is governed by the interplay between both the nuclear force and the electromagnetic force.
Visual representation of alpha decay
https://en.wikipedia.org/wiki/Beta_decay
Retrieved: 11 September 2017
Archive: https://archive.is/XREF9
Beta decay
In nuclear physics, beta decay (ß-decay) is a type of radioactive decay in which a beta ray (fast energetic electron or positron) and a neutrino are emitted from an atomic nucleus. For example, beta decay of a neutron transforms it into a proton by the emission of an electron, or conversely a proton is converted into a neutron by the emission of a positron (positron emission), thus changing the nuclide type. Neither the beta particle nor its associated neutrino exist within the nucleus prior to beta decay, but are created in the decay process. By this process, unstable atoms obtain a more stable ratio of protons to neutrons. The probability of a nuclide decaying due to beta and other forms of decay is determined by its nuclear binding energy. The binding energies of all existing nuclides form what is called the nuclear valley of stability.[1] For either electron or positron emission to be energetically possible, the energy release (see below) or Q value must be positive.
Beta decay is a consequence of the weak force, which is characterized by relatively lengthy decay times. Nucleons are composed of up or down quarks,[2] and the weak force allows a quark to change type by the exchange of a W boson and the creation of an electron/antineutrino or positron/neutrino pair. For example, a neutron, composed of two down quarks and an up quark, decays to a proton composed of a down quark and two up quarks. Decay times for many nuclides that are subject to beta decay can be thousands of years.
Electron capture is sometimes included as a type of beta decay,[3] because the basic nuclear process, mediated by the weak force, is the same. In electron capture, an inner atomic electron is captured by a proton in the nucleus, transforming it into a neutron, and an electron neutrino is released.
ß- decay in an atomic nucleus (the accompanying antineutrino is omitted). The inset shows beta decay of a free neutron. In both processes, an intermediate virtual W- boson is not shown.
https://en.wikipedia.org/wiki/Nuclear_reaction
Retrieved: 8 September 2017
Archive: https://archive.is/Puls6
Nuclear reaction
In nuclear physics and nuclear chemistry, a nuclear reaction is semantically considered to be the process in which two nuclei, or else a nucleus of an atom and a subatomic particle (such as a proton, neutron, or high energy electron) from outside the atom, collide to produce one or more nuclides that are different from the nuclide(s) that began the process. Thus, a nuclear reaction must cause a transformation of at least one nuclide to another. If a nucleus interacts with another nucleus or particle and they then separate without changing the nature of any nuclide, the process is simply referred to as a type of nuclear scattering, rather than a nuclear reaction.
In principle, a reaction can involve more than two particles colliding, but because the probability of three or more nuclei to meet at the same time at the same place is much less than for two nuclei, such an event is exceptionally rare (see triple alpha process for an example very close to a three-body nuclear reaction). "Nuclear reaction" is a term implying an induced change in a nuclide, and thus it does not apply to any type of radioactive decay (which by definition is a spontaneous process).
Natural nuclear reactions occur in the interaction between cosmic rays and matter, and nuclear reactions can be employed artificially to obtain nuclear energy, at an adjustable rate, on demand. Perhaps the most notable nuclear reactions are the nuclear chain reactions in fissionable materials that produce induced nuclear fission, and the various nuclear fusion reactions of light elements that power the energy production of the Sun and stars.

…
…
History
In 1919, Ernest Rutherford was able to accomplish transmutation of nitrogen into oxygen at the University of Manchester, using alpha particles directed at nitrogen 14N + a ? 16O + p. This was the first observation of an induced nuclear reaction, that is, a reaction in which particles from one decay are used to transform another atomic nucleus. Eventually, in 1932 at Cambridge University, a fully artificial nuclear reaction and nuclear transmutation was achieved by Rutherford's colleagues John Cockcroft and Ernest Walton, who used artificially accelerated protons against lithium-7, to split the nucleus into two alpha particles. The feat was popularly known as "splitting the atom", although it was not the modern nuclear fission reaction later discovered in heavy elements, in 1938 by the German scientists Otto Hahn and Fritz Strassmann.[3]
…
Reactions with neutrons
Reactions with neutrons are important in nuclear reactors and nuclear weapons.
https://en.wikipedia.org/wiki/Nuclear_chain_reaction
Retrieved: 15 September 2017
Archive: https://archive.is/mNFsV
Nuclear chain reaction
A nuclear chain reaction occurs when one single nuclear reaction causes an average of one or more subsequent nuclear reactions, thus leading to the possibility of a self-propagating series of these reactions. The specific nuclear reaction may be the fission of heavy isotopes (e.g., uranium-235, 235U). The nuclear chain reaction releases several million times more energy per reaction than any chemical reaction.
A possible nuclear fission chain reaction. 1. A uranium-235 atom absorbs a neutron, and fissions into two new atoms (fission fragments), releasing three new neutrons and a large amount of binding energy. 2. One of those neutrons is absorbed by an atom of uranium-238, and does not continue the reaction. Another neutron leaves the system without being absorbed. However, one neutron does collide with an atom of uranium-235, which then fissions and releases two neutrons and more binding energy. 3. Both of those neutrons collide with uranium-235 atoms, each of which fissions and releases a few neutrons, which can then continue the reaction.
https://en.wikipedia.org/wiki/Nuclide
Retrieved: 14 September 2017
Archive: https://archive.is/xmZX3
Nuclide
A nuclide (from nucleus) is an atomic species characterized by the specific constitution of its nucleus, i.e., by its number of protons Z, its number of neutrons N, and its nuclear energy state.[1]
The word nuclide was proposed[2] by Truman P. Kohman[3] in 1947. Kohman originally suggested nuclide as referring to a "species of nucleus" defined by containing a certain number of neutrons and protons. The word thus was originally intended to focus on the nucleus.
Nuclides vs isotopes[edit]
Nuclide refers to a nucleus rather than to an atom.
…
The nuclide concept (referring to individual nuclear species) emphasizes nuclear properties over chemical properties, while the isotope concept (grouping all atoms of each element) emphasizes chemical over nuclear. The neutron number has large effects on nuclear properties, but its effect on chemical properties is negligible for most elements. Even in the case of the very lightest elements where the ratio of neutron number to atomic number varies the most between isotopes it usually has only a small effect, although it does matter in some circumstances (for hydrogen, the lightest element, the isotope effect is large enough to strongly affect biology). Since isotope is the older term, it is better known than nuclide, and is still sometimes used in contexts where nuclide might be more appropriate, such as nuclear technology and nuclear medicine.
…
https://en.wikipedia.org/wiki/Mass_number
Retrieved: 14 September 2017
Archive: https://archive.is/CRgCM
Mass number
The mass number (symbol A), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It determines the atomic mass of atoms. Because protons and neutrons both are baryons, the mass number A is identical with the baryon number B as of the nucleus as of the whole atom or ion.
https://en.wikipedia.org/wiki/Table_of_nuclides
Retrieved: 8 September 2017
Archive: https://archive.is/nTnQP
Table of nuclides
A table of nuclides or chart of nuclides is a two-dimensional graph in which one axis represents the number of neutrons and the other represents the number of protons in an atomic nucleus. Each point plotted on the graph thus represents the nuclide of a real or hypothetical chemical element. This system of ordering nuclides can offer a greater insight into the characteristics of isotopes than the better-known periodic table, which shows only elements instead of each of their isotopes.
…
Full table
The isotope table below shows isotopes of the chemical elements, including all with half-life of at least one day.[3] They are arranged with increasing atomic numbers from left to right and increasing neutron numbers from top to bottom.
Cell colour denotes the half-life of each isotope; if a border is present, its colour indicates the half-life of the most stable nuclear isomer. In graphical browsers, each isotope also has a tool tip indicating its half-life. Each color represents certain range of length of half-life, and the color of border indicates the half-life of its nuclear isomer state. Some nuclides have multiple nuclear isomers, and this table notes the longest one. Dotted borders mean that a nuclide has a nuclear isomer, and their color is represented the same way as for their normal counterparts.
…
Fragment of table of nuclides for polonium, radium, copernicium and curium, as seen on a monument in front of University of Warsaw's Centre of New Technologies
https://en.wikipedia.org/wiki/Stable_nuclide
Retrieved: 14 September 2017
Archive: https://archive.is/TA2cd
Stable nuclide
Stable nuclides are nuclides that are not radioactive and so (unlike radionuclides) do not spontaneously undergo radioactive decay. When such nuclides are referred to in relation to specific elements, they are usually termed stable isotopes.
The 80 elements with one or more stable isotopes comprise a total of 253 nuclides that have not been known to decay using current equipment (see list at the end of this article). Of these elements, 26 have only one stable isotope; they are thus termed monoisotopic. The rest have more than one stable isotope. Tin has ten stable isotopes, the largest number known for an element.
Graph of nuclides (isotopes) by type of decay. Orange and blue nuclides are unstable, with the black squares between these regions representing stable nuclides. The unbroken line passing below many of the nuclides represents the theoretical position on the graph of nuclides for which proton number is the same as neutron number. The graph shows that elements with more than 20 protons must have more neutrons than protons in order to be stable.
https://en.wikipedia.org/wiki/Deuterium
Retrieved: 8 September 2017
Archive: https://archive.is/FnQBW
Deuterium
Deuterium (symbol D or 2H, also known as heavy hydrogen) is one of two stable isotopes of hydrogen. The nucleus of deuterium, called a deuteron, contains one proton and one neutron, whereas the far more common hydrogen isotope, protium, has no neutron in the nucleus. Deuterium has a natural abundance in Earth's oceans of about one atom in 6420 of hydrogen. Thus deuterium accounts for approximately 0.0156% (or on a mass basis 0.0312%) of all the naturally occurring hydrogen in the oceans, while the most common isotope (hydrogen-1 or protium) accounts for more than 99.98%.
https://en.wikipedia.org/wiki/Tritium
Retrieved: 12 September 2017
Archive: https://archive.is/MyoeG
Tritium
Tritium (/'tr?ti?m/ or /'tr??i?m/; symbol T or 3H, also known as hydrogen-3) is a radioactive isotope of hydrogen. The nucleus of tritium (sometimes called a triton) contains one proton and two neutrons, whereas the nucleus of protium (by far the most abundant hydrogen isotope) contains one proton and no neutrons. Naturally occurring tritium is extremely rare on Earth, where trace amounts are formed by the interaction of the atmosphere with cosmic rays. It can be produced by irradiating lithium metal or lithium bearing ceramic pebbles in a nuclear reactor. Tritium is used as a radioactive tracer, in radioluminescent light sources for watches and instruments, and, along with deuterium, as a fuel for nuclear fusion reactions with applications in energy generation and weapons. The name of this isotope is derived from Greek t??t?? (trítos), meaning 'third'.
Hydrogen-3 or Tritium The table is a simplified version of w:Table of nuclides (complete). It contains isotopes arranged by proton number from left to right and by neutron number from top to down, where blue symbolizes stable isotopes and orange symbolizes unstable isotopes.
https://en.wikipedia.org/wiki/Radioactive_tracer
Retrieved: 12 September 2017
Archive: https://archive.is/iUdBY
Radioactive tracer
A radioactive tracer, or radioactive label, is a chemical compound in which one or more atoms have been replaced by a radioisotope so by virtue of its radioactive decay it can be used to explore the mechanism of chemical reactions by tracing the path that the radioisotope follows from reactants to products. Radiolabeling is thus the radioactive form of isotopic labeling.
https://en.wikipedia.org/wiki/Radionuclide
Retrieved: 12 September 2017
Archive: https://archive.is/WOK4T
Radionuclide
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is an atom that has excess nuclear energy, making it unstable. This excess energy can be either emitted from the nucleus as gamma radiation, or create and emit from the nucleus a new particle (alpha particle or beta particle), or transfer this excess energy to one of its electrons, causing that electron to be ejected as a conversion electron. During those processes, the radionuclide is said to undergo radioactive decay.[1] These emissions constitute ionizing radiation. The unstable nucleus is more stable following the emission, but will sometimes undergo further decay. Radioactive decay is a random process at the level of single atoms: it is impossible to predict when one particular atom will decay.[2][3][4][5] However, for a collection of atoms of a single element the decay rate, and thus the half-life (t1/2) for that collection can be calculated from their measured decay constants. The range of the half-lives of radioactive atoms have no known limits and span a time range of over 55 orders of magnitude.
…
All chemical elements have radionuclides. Even the lightest element, hydrogen, has a well-known radionuclide, tritium. Elements heavier than lead, and the elements technetium and promethium, exist only as radionuclides.
https://en.wikipedia.org/wiki/Radioluminescence
Retrieved: 12 September 2017
Archive: https://archive.is/LtXKT
Radioluminescence
Radioluminescence is the phenomenon by which light is produced in a material by bombardment with ionizing radiation such as alpha particles, beta particles, or gamma rays. Radioluminescence is used as a low level light source for night illumination of instruments or signage or other applications where light must be produced for long periods without external energy sources. Radioluminescent paint used to be used for clock hands and instrument dials, enabling them to be read in the dark. Radioluminescence is also sometimes seen around high-power radiation sources, such as nuclear reactors and radioisotopes.
Radioluminescent 1.8-curie (67 GBq) 6-by-0.2-inch (152.4 mm × 5.1 mm) tritium vial used as a light source. It consists of a sealed glass tube containing radioactive tritium gas whose inner surfaces are coated with a phosphor.
…
Tritium[edit]
Main article: Tritium radioluminescenceThe latest generation of radioluminescent materials is based on tritium, a radioactive isotope of hydrogen with half-life of 12.32 years that emits very low-energy beta radiation. It is used on wristwatch faces, gun sights, and emergency exit signs. The tritium gas is contained in a small glass tube, coated with a phosphor on the inside. Beta particles emitted by the tritium strike the phosphor coating and cause it to fluoresce, emitting light, usually yellow-green.
Tritium is used because it is believed to pose a negligible threat to human health, in contrast to the previous radioluminescent source, radium (below), which proved to be a significant radiological hazard. The low-energy 5.7 keV beta particles emitted by tritium cannot pass through the enclosing glass tube. Even if they could, they are not able to penetrate human skin. Tritium is only a health threat if ingested. Since tritium is a gas, if a tritium tube breaks, the gas dissipates in the air and is diluted to safe concentrations. Tritium has a half-life of 12.3 years, so the brightness of a tritium light source will decline to half its initial value in that time.
Watch face illuminated by tritium tubes
…
A 1950s radium clock, exposed to ultraviolet light to increase luminescence
https://en.wikipedia.org/wiki/Nuclear_fission
Retrieved: 8 September 2017
Archive: https://archive.is/056om
Nuclear fission
In nuclear physics and nuclear chemistry, nuclear fission is either a nuclear reaction or a radioactive decay process in which the nucleus of an atom splits into smaller parts (lighter nuclei). The fission process often produces free neutrons and gamma photons, and releases a very large amount of energy even by the energetic standards of radioactive decay.
Nuclear fission of heavy elements was discovered on December 17, 1938 by German Otto Hahn and his assistant Fritz Strassmann, and explained theoretically in January 1939 by Lise Meitner and her nephew Otto Robert Frisch. Frisch named the process by analogy with biological fission of living cells. It is an exothermic reaction which can release large amounts of energy both as electromagnetic radiation and as kinetic energy of the fragments (heating the bulk material where fission takes place). In order for fission to produce energy, the total binding energy of the resulting elements must be less negative (higher energy) than that of the starting element.
Fission is a form of nuclear transmutation because the resulting fragments are not the same element as the original atom. The two nuclei produced are most often of comparable but slightly different sizes, typically with a mass ratio of products of about 3 to 2, for common fissile isotopes.[1][2] Most fissions are binary fissions (producing two charged fragments), but occasionally (2 to 4 times per 1000 events), three positively charged fragments are produced, in a ternary fission. The smallest of these fragments in ternary processes ranges in size from a proton to an argon nucleus.
Apart from fission induced by a neutron, harnessed and exploited by humans, a natural form of spontaneous radioactive decay (not requiring a neutron) is also referred to as fission, and occurs especially in very high-mass-number isotopes. Spontaneous fission was discovered in 1940 by Flyorov, Petrzhak and Kurchatov[3] in Moscow, when they decided to confirm that, without bombardment by neutrons, the fission rate of uranium was indeed negligible, as predicted by Niels Bohr; it was not.[3]
The unpredictable composition of the products (which vary in a broad probabilistic and somewhat chaotic manner) distinguishes fission from purely quantum-tunneling processes such as proton emission, alpha decay, and cluster decay, which give the same products each time. Nuclear fission produces energy for nuclear power and drives the explosion of nuclear weapons. Both uses are possible because certain substances called nuclear fuels undergo fission when struck by fission neutrons, and in turn emit neutrons when they break apart. This makes possible a self-sustaining nuclear chain reaction that releases energy at a controlled rate in a nuclear reactor or at a very rapid uncontrolled rate in a nuclear weapon.
The amount of free energy contained in nuclear fuel is millions of times the amount of free energy contained in a similar mass of chemical fuel such as gasoline, making nuclear fission a very dense source of energy. The products of nuclear fission, however, are on average far more radioactive than the heavy elements which are normally fissioned as fuel, and remain so for significant amounts of time, giving rise to a nuclear waste problem. Concerns over nuclear waste accumulation and over the destructive potential of nuclear weapons are a counterbalance to the peaceful desire to use fission as an energy source, and give rise to ongoing political debate over nuclear power.
An induced fission reaction. A neutron is absorbed by a uranium-235 nucleus, turning it briefly into an excited uranium-236 nucleus, with the excitation energy provided by the kinetic energy of the neutron plus the forces that bind the neutron. The uranium-236, in turn, splits into fast-moving lighter elements (fission products) and releases three free neutrons. At the same time, one or more "prompt gamma rays" (not shown) are produced, as well.
A visual representation of an induced nuclear fission event where a slow-moving neutron is absorbed by the nucleus of a uranium-235 atom, which fissions into two fast-moving lighter elements (fission products) and additional neutrons. Most of the energy released is in the form of the kinetic velocities of the fission products and the neutrons.
Here is a link to the gif.
…
The mushroom cloud of the atomic bomb dropped on Nagasaki, Japan on August 9, 1945, rose over 18 kilometres (11 mi) above the bomb's hypocenter. An estimated 39,000 people were killed by the atomic bomb,[11] of whom 23,145–28,113 were Japanese factory workers, 2,000 were Korean slave laborers, and 150 were Japanese combatants.[12][13][14]
https://en.wikipedia.org/wiki/Nuclear_transmutation
Retrieved: 11 September 2017
Archive: https://archive.is/QAtgm
Nuclear transmutation
Nuclear transmutation is the conversion of one chemical element or an isotope into another.[1] Because any element (or isotope of one) is defined by its number of protons (and neutrons) in its atoms, i.e. in the atomic nucleus, nuclear transmutation occurs in any process where the number of protons or neutrons in the nucleus is changed.
A transmutation can be achieved either by nuclear reactions (in which an outside particle reacts with a nucleus) or by radioactive decay where no outside cause is needed.
Natural transmutation by stellar nucleosynthesis in the past created most of the heavier chemical elements in the known existing universe, and continues to take place to this day, creating the vast majority of the most common elements in the universe, including helium, oxygen and carbon. Most stars carry out transmutation through fusion reactions involving Hydrogen and helium, while much larger stars are also capable of fusing heavier elements up to iron late in their evolution.
Elements heavier than iron, such as gold and lead, are created through elemental transmutations that can only take place in supernovae - as stars begin to fuse heavier elements, substantially less energy is released from each fusion reaction, and each fusion reaction that produces elements heavier than iron is endothermic in nature, and stars are incapable of carrying this out.
The Sun is a natural fusion reactor, and transmutates light elements into heavier elements through stellar nucleosynthesis, a form of nuclear fusion.
https://en.wikipedia.org/wiki/Nuclear_fusion
Retrieved: 11 September 2017
Archive: https://archive.is/8uEOA
Nuclear fusion
In nuclear physics, nuclear fusion is a reaction in which two or more atomic nuclei come close enough to form one or more different atomic nuclei and subatomic particles (neutrons or protons). The difference in mass between the products and reactants is manifested as the release of large amounts of energy. This difference in mass arises due to the difference in atomic "binding energy" between the atomic nuclei before and after the reaction. Fusion is the process that powers active or "main sequence" stars, or other high magnitude stars.
The fusion process that produces a nucleus lighter than iron-56 or nickel-62 will generally yield a net energy release. These elements have the smallest mass per nucleon and the largest binding energy per nucleon, respectively. Fusion of light elements toward these releases energy (an exothermic process), while a fusion producing nuclei heavier than these elements, will result in energy retained by the resulting nucleons, and the resulting reaction is endothermic. The opposite is true for the reverse process, nuclear fission. This means that the lighter elements, such as hydrogen and helium, are in general more fusible; while the heavier elements, such as uranium and plutonium, are more fissionable. The extreme astrophysical event of a supernova can produce enough energy to fuse nuclei into elements heavier than iron.
Following the discovery of quantum tunneling by physicist Friedrich Hund, in 1929 Robert Atkinson and Fritz Houtermans used the measured masses of light elements to predict that large amounts of energy could be released by fusing small nuclei. Building upon the nuclear transmutation experiments by Ernest Rutherford, carried out several years earlier, the laboratory fusion of hydrogen isotopes was first accomplished by Mark Oliphant in 1932. During the remainder of that decade the steps of the main cycle of nuclear fusion in stars were worked out by Hans Bethe. Research into fusion for military purposes began in the early 1940s as part of the Manhattan Project. Fusion was accomplished in 1951 with the Greenhouse Item nuclear test. Nuclear fusion on a large scale in an explosion was first carried out on November 1, 1952, in the Ivy Mike hydrogen bomb test.
Research into developing controlled thermonuclear fusion for civil purposes also began in earnest in the 1950s, and it continues to this day.
The Sun is a main-sequence star, and thus generates its energy by nuclear fusion of hydrogen nuclei into helium. In its core, the Sun fuses 4 million metric tons of hydrogen each second.
…
Fusion of deuterium with tritium creating helium-4, freeing a neutron, and releasing 17.59 MeV as kinetic energy of the products while a corresponding amount of mass disappears, in agreement with kinetic E= ?mc2, where ?m is the decrease in the total rest mass of particles.[1]
…
The electrostatic force between the positively charged nuclei is repulsive, but when the separation is small enough, the quantum effect will tunnel through the wall. Therefore, the prerequisite for fusion is that the two nuclei be brought close enough together for a long enough time for quantum tunnelling to act.
…
The only man-made fusion device to achieve ignition to date is the hydrogen bomb.[citation needed] The detonation of the first device, codenamed Ivy Mike, occurred in 1952 and is shown here.
https://en.wikipedia.org/wiki/Quantum_tunnelling
Retrieved: 11 September 2017
Archive: https://archive.is/S5VMl
Quantum tunnelling
Quantum tunnelling or tunneling (see spelling differences) refers to the quantum mechanical phenomenon where a particle tunnels through a barrier that it classically could not surmount. This plays an essential role in several physical phenomena, such as the nuclear fusion that occurs in main sequence stars like the Sun.[1] It has important applications to modern devices such as the tunnel diode,[2] quantum computing, and the scanning tunnelling microscope. The effect was predicted in the early 20th century and its acceptance as a general physical phenomenon came mid-century.[3]
Fundamental quantum mechanical concepts are central to this phenomenon, which makes quantum tunnelling one of the novel implications of quantum mechanics. Quantum tunneling is projected to create physical limits to how small transistors can get, due to electrons being able to tunnel past them if they are too small.[citation needed]
Tunnelling is often explained in terms of Heisenberg uncertainty principle and the wave–particle duality of matter.
…
Introduction to the concept
Quantum tunnelling falls under the domain of quantum mechanics: the study of what happens at the quantum scale. This process cannot be directly perceived, but much of its understanding is shaped by the microscopic world, which classical mechanics cannot adequately explain. To understand the phenomenon, particles attempting to travel between potential barriers can be compared to a ball trying to roll over a hill; quantum mechanics and classical mechanics differ in their treatment of this scenario. Classical mechanics predicts that particles that do not have enough energy to classically surmount a barrier will not be able to reach the other side. Thus, a ball without sufficient energy to surmount the hill would roll back down. Or, lacking the energy to penetrate a wall, it would bounce back (reflection) or in the extreme case, bury itself inside the wall (absorption). In quantum mechanics, these particles can, with a very small probability, tunnel to the other side, thus crossing the barrier. Here, the "ball" could, in a sense, borrow energy from its surroundings to tunnel through the wall or "roll over the hill", paying it back by making the reflected electrons more energetic than they otherwise would have been.[11]
The reason for this difference comes from the treatment of matter in quantum mechanics as having properties of waves and particles. One interpretation of this duality involves the Heisenberg uncertainty principle, which defines a limit on how precisely the position and the momentum of a particle can be known at the same time.[4] This implies that there are no solutions with a probability of exactly zero (or one), though a solution may approach infinity if, for example, the calculation for its position was taken as a probability of 1, the other, i.e. its speed, would have to be infinity. Hence, the probability of a given particle's existence on the opposite side of an intervening barrier is non-zero, and such particles will appear on the 'other' (a semantically difficult word in this instance) side with a relative frequency proportional to this probability.
Retrieved: 11 September 2017
Archive: https://archive.is/SEIUm
Very interesting animation!
https://en.wikipedia.org/wiki/Nuclear_weapon
Retrieved: 11 September 2017
Archive: https://archive.is/6w7u7
Nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or from a combination of fission and fusion reactions (thermonuclear bomb). Both bomb types release large quantities of energy from relatively small amounts of matter. The first test of a fission ("atomic") bomb released an amount of energy approximately equal to 20,000 tons of TNT (84 TJ). The first thermonuclear ("hydrogen") bomb test released energy approximately equal to 10 million tons of TNT (42 PJ).[1] A thermonuclear weapon weighing little more than 2,400 pounds (1,100 kg) can release energy equal to more than 1.2 million tons of TNT (5.0 PJ).[2] A nuclear device no larger than traditional bombs can devastate an entire city by blast, fire, and radiation. Since they are weapons of mass destruction, the proliferation of nuclear weapons is a focus of international relations policy.
Nuclear weapons have been used twice in war, both times by the United States against Japan near the end of World War II. On August 6, 1945, the U.S. Army Air Forces detonated a uranium gun-type fission bomb nicknamed "Little Boy" over the Japanese city of Hiroshima; three days later, on August 9, the U.S. Army Air Forces detonated a plutonium implosion-type fission bomb nicknamed "Fat Man" over the Japanese city of Nagasaki. These bombings resulted in the deaths of approximately 200,000 civilians and military personnel from injuries sustained from the explosions.[3] The ethics of these bombings and their role in Japan's surrender are subjects of debate.
MES Note: Given 9/11 and PizzaGate, calling atomic bombs that killed thousands "Fat Man" and "Little Boy" could not have been more disturbingly fitting… # YouCantMakeThisStuffUp
Since the atomic bombings of Hiroshima and Nagasaki, nuclear weapons have been detonated over two thousand times for testing and demonstration. Only a few nations possess such weapons or are suspected of seeking them. The only countries known to have detonated nuclear weapons—and acknowledge possessing them—are (chronologically by date of first test) the United States, the Soviet Union (succeeded as a nuclear power by Russia), the United Kingdom, France, China, India, Pakistan, and North Korea. Israel is believed to possess nuclear weapons, though, in a policy of deliberate ambiguity, it does not acknowledge having them. Germany, Italy, Turkey, Belgium and the Netherlands are nuclear weapons sharing states.[4][5][6] South Africa is the only country to have independently developed and then renounced and dismantled its nuclear weapons.[7]
The Treaty on the Non-Proliferation of Nuclear Weapons aims to reduce the spread of nuclear weapons, but its effectiveness has been questioned, and political tensions remained high in the 1970s and 1980s. Modernisation of weapons continues to this day.[8]
The mushroom cloud of the atomic bombing of the Japanese city of Nagasaki on August 9, 1945 rose some 11 mi (18 km) above the bomb's hypocenter.
A mockup of the Fat Man nuclear device.
…
Types
There are two basic types of nuclear weapons: those that derive the majority of their energy from nuclear fission reactions alone, and those that use fission reactions to begin nuclear fusion reactions that produce a large amount of the total energy output.[9]
Fission weapons
All existing nuclear weapons derive some of their explosive energy from nuclear fission reactions. Weapons whose explosive output is exclusively from fission reactions are commonly referred to as atomic bombs or atom bombs (abbreviated as A-bombs). This has long been noted as something of a misnomer, as their energy comes from the nucleus of the atom, just as it does with fusion weapons.
In fission weapons, a mass of fissile material (enriched uranium or plutonium) is forced into supercriticality—allowing an exponential growth of nuclear chain reactions—either by shooting one piece of sub-critical material into another (the "gun" method) or by compressing using explosive lenses a sub-critical sphere of material using chemical explosives to many times its original density (the "implosion" method). The latter approach is considered more sophisticated than the former, and only the latter approach can be used if the fissile material is plutonium.[10]
A major challenge in all nuclear weapon designs is to ensure that a significant fraction of the fuel is consumed before the weapon destroys itself. The amount of energy released by fission bombs can range from the equivalent of just under a ton to upwards of 500,000 tons (500 kilotons) of TNT (4.2 to 2.1×108 GJ).[11]
All fission reactions generate fission products, the remains of the split atomic nuclei. Many fission products are either highly radioactive (but short-lived) or moderately radioactive (but long-lived), and as such, they are a serious form of radioactive contamination if not fully contained. Fission products are the principal radioactive component of nuclear fallout.
The most commonly used fissile materials for nuclear weapons applications have been uranium-235 and plutonium-239. Less commonly used has been uranium-233. Neptunium-237 and some isotopes of americium may be usable for nuclear explosives as well, but it is not clear that this has ever been implemented, and their plausible use in nuclear weapons is a matter of dispute.[12]
The two basic fission weapon designs
Fusion weapons
Main article: Thermonuclear weaponThe other basic type of nuclear weapon produces a large proportion of its energy in nuclear fusion reactions. Such fusion weapons are generally referred to as thermonuclear weapons or more colloquially as hydrogen bombs (abbreviated as H-bombs), as they rely on fusion reactions between isotopes of hydrogen (deuterium and tritium). All such weapons derive a significant portion of their energy from fission reactions used to "trigger" fusion reactions, and fusion reactions can themselves trigger additional fission reactions.[13]
Only six countries—United States, Russia, United Kingdom, China, France, and India—have conducted thermonuclear weapon tests. (Whether India has detonated a "true" multi-staged thermonuclear weapon is controversial.)[14] North Korea claims to have tested a fusion weapon as of January 2016, though this claim is disputed.[15] Thermonuclear weapons are considered much more difficult to successfully design and execute than primitive fission weapons. Almost all of the nuclear weapons deployed today use the thermonuclear design because it is more efficient.[citation needed]
Thermonuclear bombs work by using the energy of a fission bomb to compress and heat fusion fuel. In the Teller-Ulam design, which accounts for all multi-megaton yield hydrogen bombs, this is accomplished by placing a fission bomb and fusion fuel (tritium, deuterium, or lithium deuteride) in proximity within a special, radiation-reflecting container. When the fission bomb is detonated, gamma rays and X-rays emitted first compress the fusion fuel, then heat it to thermonuclear temperatures. The ensuing fusion reaction creates enormous numbers of high-speed neutrons, which can then induce fission in materials not normally prone to it, such as depleted uranium. Each of these components is known as a "stage", with the fission bomb as the "primary" and the fusion capsule as the "secondary". In large, megaton-range hydrogen bombs, about half of the yield comes from the final fissioning of depleted uranium.[11
Virtually all thermonuclear weapons deployed today use the "two-stage" design described above, but it is possible to add additional fusion stages—each stage igniting a larger amount of fusion fuel in the next stage. This technique can be used to construct thermonuclear weapons of arbitrarily large yield, in contrast to fission bombs, which are limited in their explosive force. The largest nuclear weapon ever detonated, the Tsar Bomba of the USSR, which released an energy equivalent of over 50 megatons of TNT (210 PJ), was a three-stage weapon. Most thermonuclear weapons are considerably smaller than this, due to practical constraints from missile warhead space and weight requirements.[16]
Fusion reactions do not create fission products, and thus contribute far less to the creation of nuclear fallout than fission reactions, but because all thermonuclear weapons contain at least one fission stage, and many high-yield thermonuclear devices have a final fission stage, thermonuclear weapons can generate at least as much nuclear fallout as fission-only weapons.
The basics of the Teller–Ulam design for a hydrogen bomb: a fission bomb uses radiation to compress and heat a separate section of fusion fuel.
…
A demilitarized, commercial launch of the Russian Strategic Rocket Forces R-36 ICBM; also known by the NATO reporting name: SS-18 Satan. Upon its first fielding in the late 1960s, the SS-18 remains the single highest throw weight missile delivery system ever built.
…
Montage of an inert test of a United States Trident SLBM (submarine launched ballistic missile), from submerged to the terminal, or re-entry phase, of the multiple independently targetable reentry vehicles
…
The now decommissioned United States' Peacekeeper missile was an ICBM developed to replace the Minuteman missile in the late 1980s. Each missile, like the heavier lift Russian SS-18 Satan, could contain up to ten nuclear warheads (shown in red), each of which could be aimed at a different target. A factor in the development of MIRVs was to make complete missile defense difficult for an enemy country.
…
The International Atomic Energy Agency was created in 1957 to encourage peaceful development of nuclear technology while providing international safeguards against nuclear proliferation.
…
The USSR and United States nuclear weapon stockpiles throughout the Cold War until 2015, with a precipitous drop in total numbers following the end of the Cold War in 1991.
…
Over 2,000 nuclear tests have been conducted in over a dozen different sites around the world. Red Russia/Soviet Union, blue France, light blue United States, violet Britain, black Israel, yellow China, orange India, brown Pakistan, green North Korea and light green (territories exposed to nuclear bombs)
…
This view of downtown Las Vegas shows a mushroom cloud in the background. Scenes such as this were typical during the 1950s. From 1951 to 1962 the government conducted 100 atmospheric tests at the nearby Nevada Test Site.
…
Effects of nuclear explosions on human health
Main article: Effects of nuclear explosions on human healthSome scientists estimate that a nuclear war with 100 Hiroshima-size nuclear explosions on cities could cost the lives of tens of millions of people from long term climatic effects alone. The climatology hypothesis is that if each city firestorms, a great deal of soot could be thrown up into the atmosphere which could blanket the earth, cutting out sunlight for years on end, causing the disruption of food chains, in what is termed a nuclear winter.[72][73]
People near the Hiroshima explosion and who managed to survive the explosion subsequently suffered a variety of medical effects:[74][75][citation needed][76]
• Initial stage—the first 1–9 weeks, in which are the greatest number of deaths, with 90% due to thermal injury and/or blast effects and 10% due to super-lethal radiation exposure.
• Intermediate stage—from 10–12 weeks. The deaths in this period are from ionizing radiation in the median lethal range – LD50
• Late period—lasting from 13–20 weeks. This period has some improvement in survivors' condition.
• Delayed period—from 20+ weeks. Characterized by numerous complications, mostly related to healing of thermal and mechanical injuries, and if the individual was exposed to a few hundred to a thousand millisieverts of radiation, it is coupled with infertility, sub-fertility and blood disorders. Furthermore, ionizing radiation above a dose of around 50–100 millisievert exposure has been shown to statistically begin increasing one's chance of dying of cancer sometime in their lifetime over the normal unexposed rate of ~25%, in the long term, a heightened rate of cancer, proportional to the dose received, would begin to be observed after ~5+ years, with lesser problems such as eye cataracts and other more minor effects in other organs and tissue also being observed over the long term.Fallout exposure – Depending on if further afield individuals shelter in place or evacuate perpendicular to the direction of the wind, and therefore avoid contact with the fallout plume, and stay there for the days and weeks after the nuclear explosion, their exposure to fallout, and therefore their total dose, will vary. With those who do shelter in place, and or evacuate, experiencing a total dose that would be negligible in comparison to someone who just went about their life as normal.[77][78]
Staying indoors until after the most hazardous fallout isotope, I-131 decays away to 0.1% of its initial quantity after ten half lifes – which is represented by 80 days in I-131s case, would make the difference between likely contracting Thyroid cancer or escaping completely from this substance depending on the actions of the individual.[79]
https://en.wikipedia.org/wiki/Fissile_material
Retrieved: 15 September 2017
Archive: https://archive.is/kJn46
Fissile material
In nuclear engineering, fissile material is material capable of sustaining a nuclear fission chain reaction.
https://en.wikipedia.org/wiki/Critical_mass
Retrieved: 15 September 2017
Archive: https://archive.is/Rd8Hk
Critical mass
A critical mass is the smallest amount of fissile material needed for a sustained nuclear chain reaction. The critical mass of a fissionable material depends upon its nuclear properties (specifically, the nuclear fission cross-section), its density, its shape, its enrichment, its purity, its temperature, and its surroundings. The concept is important in nuclear weapon design.
Explanation of criticality
When a nuclear chain reaction in a mass of fissile material is self-sustaining, the mass is said to be in a critical state in which there is no increase or decrease in power, temperature, or neutron population.
…
A subcritical mass is a mass of fissile material that does not have the ability to sustain a fission chain reaction.
…
A supercritical mass is one where there is an increasing rate of fission.
https://en.wikipedia.org/wiki/Explosive_lens
Retrieved: 11 September 2017
Archive: https://archive.is/ki9jc
Explosive lens
An explosive lens—as used, for example, in nuclear weapons—is a highly specialized shaped charge. In general, it is a device composed of several explosive charges. These charges are arranged and formed with the intent to control the shape of the detonation wave passing through them. The explosive lens is conceptually similar to an optical lens, which focuses light waves. The charges that make up the explosive lens are chosen to have different rates of detonation. In order to convert a spherically expanding wavefront into a spherically converging one using only a single boundary between the constituent explosives, the boundary shape must be a paraboloid; similarly, to convert a spherically diverging front into a flat one, the boundary shape must be a hyperboloid, and so on. Several boundaries can be used to reduce aberrations (deviations from intended shape) of the final wavefront.
Modern high explosive lenses. The colored areas are the fast explosive, while the white areas are the slow explosives.
In an implosion-type nuclear weapon, polygonal lenses are arranged around the spherical core of the bomb. Thirty-two "points" are shown. Other designs use as many as 96 or as few as two such points.
Use in nuclear weapons
In a nuclear weapon, an array of explosive lenses is used to change the several approximately-spherical diverging detonation waves into a single spherical converging one. The converging wave is then used to collapse the various shells (tamper, reflector, pusher, etc.) and finally compresses the core (pit) of fissionable material to a prompt critical state.
Cross-section of the "Trinity" gadget. The alternating high and slow explosives (in purple) are the explosive lens which forces the spherical core to compress into prompt criticality.
…
Other uses
Lenses using alternate design techniques and producing flat "plane wave" outputs are used for high transient pressure physics and materials science experiments.[3]
https://en.wikipedia.org/wiki/Manhattan_Project
Retrieved: 12 September 2017
Archive: https://archive.is/qhOef
Manhattan Project
This article is about the atomic bomb project. For other uses, see Manhattan Project (disambiguation).
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project was under the direction of Major General Leslie Groves of the U.S. Army Corps of Engineers. Nuclear physicist Robert Oppenheimer was the director of the Los Alamos Laboratory that designed the actual bombs. The Army component of the project was designated the Manhattan District; "Manhattan" gradually superseded the official codename, Development of Substitute Materials, for the entire project. Along the way, the project absorbed its earlier British counterpart, Tube Alloys. The Manhattan Project began modestly in 1939, but grew to employ more than 130,000 people and cost nearly US $2 billion (about $27 billion in 2016[1] dollars). Over 90% of the cost was for building factories and to produce fissile material, with less than 10% for development and production of the weapons. Research and production took place at more than 30 sites across the United States, the United Kingdom, and Canada.
Two types of atomic bombs were developed concurrently during the war: a relatively simple gun-type fission weapon and a more complex implosion-type nuclear weapon. The Thin Man gun-type design proved impractical to use with plutonium so a simpler gun-type called Little Boy was developed that used uranium-235, an isotope that makes up only 0.7 percent of natural uranium. Chemically identical to the most common isotope, uranium-238, and with almost the same mass, it proved difficult to separate the two. Three methods were employed for uranium enrichment: electromagnetic, gaseous and thermal. Most of this work was performed at the Clinton Engineer Works at Oak Ridge, Tennessee.
In parallel with the work on uranium was an effort to produce plutonium. After the feasibility of the world's first artificial nuclear reactor was demonstrated in Chicago at the Metallurgical Laboratory, it designed the X-10 Graphite Reactor at Oak Ridge and the production reactors in Hanford, Washington, in which uranium was irradiated and transmuted into plutonium. The plutonium was then chemically separated from the uranium. The Fat Man implosion-type weapon was developed in a concerted design and development effort by the Los Alamos Laboratory.
The project was also charged with gathering intelligence on the German nuclear weapon project. Through Operation Alsos, Manhattan Project personnel served in Europe, sometimes behind enemy lines, where they gathered nuclear materials and documents, and rounded up German scientists. Despite the Manhattan Project's tight security, Soviet atomic spies successfully penetrated the program.
The first nuclear device ever detonated was an implosion-type bomb at the Trinity test, conducted at New Mexico's Alamogordo Bombing and Gunnery Range on 16 July 1945. Little Boy and Fat Man bombs were used a month later in the atomic bombings of Hiroshima and Nagasaki, respectively. In the immediate postwar years, the Manhattan Project conducted weapons testing at Bikini Atoll as part of Operation Crossroads, developed new weapons, promoted the development of the network of national laboratories, supported medical research into radiology and laid the foundations for the nuclear navy. It maintained control over American atomic weapons research and production until the formation of the United States Atomic Energy Commission in January 1947.
The Trinity test of the Manhattan Project was the first detonation of a nuclear weapon.
Origins
The discovery of nuclear fission by German chemists Otto Hahn and Fritz Strassmann in 1938, and its theoretical explanation by Lise Meitner and Otto Frisch, made the development of an atomic bomb a theoretical possibility. There were fears that a German atomic bomb project would develop one first, especially among scientists who were refugees from Nazi Germany and other fascist countries.[2] In August 1939, Hungarian-born physicists Leó Szilárd and Eugene Wigner drafted the Einstein–Szilárd letter, which warned of the potential development of "extremely powerful bombs of a new type". It urged the United States to take steps to acquire stockpiles of uranium ore and accelerate the research of Enrico Fermi and others into nuclear chain reactions. They had it signed by Albert Einstein and delivered to President Franklin D. Roosevelt. Roosevelt called on Lyman Briggs of the National Bureau of Standards to head the Advisory Committee on Uranium to investigate the issues raised by the letter. Briggs held a meeting on 21 October 1939, which was attended by Szilárd, Wigner and Edward Teller. The committee reported back to Roosevelt in November that uranium "would provide a possible source of bombs with a destructiveness vastly greater than anything now known."[3]
…
A 1940 meeting at Berkeley: Ernest O. Lawrence, Arthur H. Compton, Vannevar Bush, James B. Conant, Karl T. Compton, and Alfred L. Loomis
…
Different fission bomb assembly methods explored during the July 1942 conference
…
Manhattan Project Organization Chart, 1 May 1946
…
A selection of US and Canadian sites important to the Manhattan Project. Click on the location for more information.
…
Los Alamos
Main article: Project Y…
Because it was secret, Los Alamos was referred to as "Site Y" or "the Hill".[96] Birth certificates of babies born in Los Alamos during the war listed their place of birth as PO Box 1663 in Santa Fe.[97]
…
Map of Los Alamos site, New Mexico, 1943–45
…
…
Operators, so-called Calutron Girls, photographed by Ed Westcott, at their calutron control panels at Y-12. Gladys Owens, the woman seated in the foreground, did not know what she had been involved with until seeing this photo in a public tour of the facility fifty years later.[147]
…
An implosion-type nuclear bomb
…
Trinity
Main article: Trinity (nuclear test)Because of the complexity of an implosion-style weapon, it was decided that, despite the waste of fissile material, an initial test would be required. Groves approved the test, subject to the active material being recovered. Consideration was therefore given to a controlled fizzle, but Oppenheimer opted instead for a full-scale nuclear test, codenamed "Trinity".[215]
…
For the actual test, the weapon, nicknamed "the gadget", was hoisted to the top of a 100-foot (30 m) steel tower, as detonation at that height would give a better indication of how the weapon would behave when dropped from a bomber. Detonation in the air maximized the energy applied directly to the target, and generated less nuclear fallout. The gadget was assembled under the supervision of Norris Bradbury at the nearby McDonald Ranch House on 13 July, and precariously winched up the tower the following day.[222] Observers included Bush, Chadwick, Conant, Farrell, Fermi, Groves, Lawrence, Oppenheimer and Tolman. At 05:30 on 16 July 1945 the gadget exploded with an energy equivalent of around 20 kilotons of TNT, leaving a crater of Trinitite (radioactive glass) in the desert 250 feet (76 m) wide. The shock wave was felt over 100 miles (160 km) away, and the mushroom cloud reached 7.5 miles (12.1 km) in height. It was heard as far away as El Paso, Texas, so Groves issued a cover story about an ammunition magazine explosion at Alamogordo Field.[223][224]
…
The explosives of "the gadget" were raised to the top of the tower for the final assembly.
…
Personnel
In June 1944, the Manhattan Project employed some 129,000 workers, of whom 84,500 were construction workers, 40,500 were plant operators and 1,800 were military personnel. As construction activity fell off, the workforce declined to 100,000 a year later, but the number of military personnel increased to 5,600. Procuring the required numbers of workers, especially highly skilled workers, in competition with other vital wartime programs proved very difficult.[225] In 1943, Groves obtained a special temporary priority for labor from the War Manpower Commission. In March 1944, both the War Production Board and the War Manpower Commission gave the project their highest priority.[226]
…
Secrecy
A 1945 Life article estimated that before the Hiroshima and Nagasaki bombings "probably no more than a few dozen men in the entire country knew the full meaning of the Manhattan Project, and perhaps only a thousand others even were aware that work on atoms was involved." The magazine wrote that the more than 100,000 others employed with the project "worked like moles in the dark". Warned that disclosing the project's secrets was punishable by 10 years in prison or a $10,000 ($133,000 today[1]) fine, they saw enormous quantities of raw materials enter factories with nothing coming out, and monitored "dials and switches while behind thick concrete walls mysterious reactions took place" without knowing the purpose of their jobs.[235][236][237][238][239]
Oak Ridge security personnel considered any private party with more than seven people as suspicious, and residents—who believed that US government agents were secretly among them—avoided repeatedly inviting the same guests. Although original residents of the area could be buried in existing cemeteries, every coffin was reportedly opened for inspection.[239] Everyone, including top military officials, and their automobiles were searched when entering and exiting project facilities. One Oak Ridge worker stated that "if you got inquisitive, you were called on the carpet within two hours by government secret agents. Usually those summoned to explain were then escorted bag and baggage to the gate and ordered to keep going." Nonetheless, despite being told that their work would help end the war and perhaps all future wars,[240] not seeing or understanding the results of their often tedious duties—or even typical side effects of factory work such as smoke from smokestacks—and the war in Europe ending without the use of their work, caused serious morale problems among workers and caused many rumors to spread. One manager stated after the war:
Well it wasn't that the job was tough ... it was confusing. You see, no one knew what was being made in Oak Ridge, not even me, and a lot of the people thought they were wasting their time here. It was up to me to explain to the dissatisfied workers that they were doing a very important job. When they asked me what, I'd have to tell them it was a secret. But I almost went crazy myself trying to figure out what was going on.[238]
Another worker told of how, working in a laundry, she every day held "a special instrument" to uniforms and listened for "a clicking noise". She learned only after the war that she had been performing the important task of checking for radiation with a geiger counter. To improve morale among such workers Oak Ridge created an extensive system of intramural sports leagues, including 10 baseball teams, 81 softball teams, and 26 football teams.[238]
A billboard encouraging secrecy among Oak Ridge workers
Censorship
Voluntary censorship of atomic information began before the Manhattan Project. After the start of the European war in 1939 American scientists began avoiding publishing military-related research, and in 1940 scientific journals began asking the National Academy of Sciences to clear articles. William L. Laurence of The New York Times, who wrote an article for The Saturday Evening Post in September 1940 on atomic fission, later learned that government officials asked librarians nationwide in 1943 to withdraw the issue.[241] The Soviets noticed the silence, however. In April 1942 nuclear physicist Georgy Flyorov wrote to Josef Stalin on the absence of articles on nuclear fission in American journals; this resulted in the Soviet Union establishing its own atomic bomb project.[242]
The Manhattan Project operated under tight security lest its discovery induce Axis powers, especially Germany, to accelerate their own nuclear projects or undertake covert operations against the project.[243] The government's Office of Censorship, by contrast, relied on the press to comply with a voluntary code of conduct it published, and the project at first avoided notifying the office. By early 1943 newspapers began publishing reports of large construction in Tennessee and Washington based on public records, and the office began discussing with the project how to maintain secrecy. In June the Office of Censorship asked newspapers and broadcasters to avoid discussing "atom smashing, atomic energy, atomic fission, atomic splitting, or any of their equivalents. The use for military purposes of radium or radioactive materials, heavy water, high voltage discharge equipment, cyclotrons." The office also asked to avoid discussion of "polonium, uranium, ytterbium, hafnium, protactinium, radium, rhenium, thorium, deuterium"; only uranium was sensitive, but was listed with other elements to hide its importance.[244][245]
MES Note: WTF??!! The US Government had a Censorship office???!!
Also, note the government strategy of disinformation by surrounding uranium with other random substances to hide its importance.
Much like filling the internet with many bogus conspiracies to hide the few real ones, such as 9/11 #FreeEnergy Tech!
Security poster, warning office workers to close drawers and put documents in safes when not being used.
…
Little Boy explodes over Hiroshima, Japan, 6 August 1945 (left);
Fat Man explodes over Nagasaki, Japan, 9 August 1945 (right).…
After the war
Seeing the work they had not understood produce the Hiroshima and Nagasaki bombs amazed the workers of the Manhattan Project as much as the rest of the world; newspapers in Oak Ridge announcing the Hiroshima bomb sold for $1 ($13 today[1]).[236][245] Although the bombs' existence was public, secrecy continued, and many workers remained ignorant of their jobs; one stated in 1946, "I don't know what the hell I'm doing besides looking into a ——— and turning a ——— alongside a ———. I don't know anything about it, and there's nothing to say". Many residents continued to avoid discussion of "the stuff" in ordinary conversation despite it being the reason for their town's existence.[239]
…
Two Fat Man–type detonations were conducted at Bikini Atoll in July 1946 as part of Operation Crossroads to investigate the effect of nuclear weapons on warships.[309] Able was detonated on 1 July 1946. The more spectacular Baker was detonated underwater on 25 July 1946.[310]
…
Following a domestic debate over the permanent management of the nuclear program, the United States Atomic Energy Commission (AEC) was created by the Atomic Energy Act of 1946 to take over the functions and assets of the Manhattan Project. It established civilian control over atomic development, and separated the development, production and control of atomic weapons from the military.
…
President Harry S. Truman signs the Atomic Energy Act of 1946, establishing the United States Atomic Energy Commission.
…
Cost
The project expenditure through 1 October 1945 was $1.845 billion, equivalent to less than nine days of wartime spending, and was $2.191 billion when the AEC assumed control on 1 January 1947. Total allocation was $2.4 billion. Over 90% of the cost was for building plants and producing the fissionable materials, and less than 10% for development and production of the weapons.[316][317]
A total of four weapons (the Trinity gadget, Little Boy, Fat Man, and an unused bomb) were produced by the end of 1945, making the average cost per bomb around $500 million in 1945 dollars. By comparison, the project's total cost by the end of 1945 was about 90% of the total spent on the production of US small arms (not including ammunition) and 34% of the total spent on US tanks during the same period.[315]
…
On handing over control to the Atomic Energy Commission, Groves bid farewell to the people who had worked on the Manhattan Project:
Five years ago, the idea of Atomic Power was only a dream. You have made that dream a reality. You have seized upon the most nebulous of ideas and translated them into actualities. You have built cities where none were known before. You have constructed industrial plants of a magnitude and to a precision heretofore deemed impossible. You built the weapon which ended the War and thereby saved countless American lives. With regard to peacetime applications, you have raised the curtain on vistas of a new world.[331]
In 2014, the United States Congress passed a law providing for a national park dedicated to the history of the Manhattan Project.[332] The Manhattan Project National Historical Park was established on 10 November 2015.[333]
UNREAL! Just a small group of men knew the actual purpose of the Manhattan Project, a few thousand knew it was about atoms, and hundreds of thousands had no idea what they were involved in!
So much for the bogus denial of "large-scale conspiracies"…
Also, very ominous message by Project leader Major General Leslie Groves…
The hidden tech (atomic bomb) that ended the world war is similar to the hidden tech (9/11) that ushered in the Middle East wars! # ConnectingDots
Thus it appears that the current world government has the technology to start wars and end them too. # NWO # WakeUpPeople # BoycottEverything
Retrieved: 12 September 2017
Archive: https://archive.is/zvv3L
Make sure to watch my earlier video on CIA discrediting critical thinkers with the "Conspiracy Theorist" label; and in which I go over an epic rant about the Wikipedia/Mainstream Media/Mainstream "Science" that large-scale conspiracies are "impossible"… # NoTheyArent # ManhattanProject
Now let's see just what kind of "men" these were that likely orchestrated the Manhattan Project…
Recall from # PizzaGate Part 28 (https://mes.fm/pizzagate-voat) that the Manhattan Project, and the United Nations, was likely first hatched at the Bohemian Grove in San Francisco, where rich powerful men meet annually in secret to perform a ritual mock human sacrifice to a giant Owl statue…
……………… These are the men that run the world…
I wonder happens behind closed doors??... Actually I don't want to know!
At least now we get a glimpse into why there is so much secrecy behind global affairs…
https://en.wikipedia.org/wiki/Office_of_Censorship#Censorship_of_the_atomic_bomb
Retrieved: 15 September 2017
Archive: https://archive.is/KGvWc
Office of Censorship
The Office of Censorship was an emergency wartime agency set up on December 19, 1941 to aid in the censorship of all communications coming into and going out of the United States. It closed in November 1945.
Overview
Voluntary censorship by the American press began before the country's entry into the war after the bombing of Pearl Harbor in December 1941. After the European war began in 1939, journalists began withholding information about Canadian troop movements.[1]:21 The First War Powers Act, approved on December 18, 1941, contained broad grants of Executive authority for the prosecution of the war, including a provision for censorship. The next day President Franklin D. Roosevelt signed Executive Order 8985, which established the Office of Censorship and conferred on its director the power to censor international communications in "his absolute discretion."
…
Censorship of the atomic bomb[edit]
Price called the Manhattan Project, the United States' development of the atomic bomb, the best-kept secret of the war.[3] It and radar were the two military topics that, if a code violation occurred, his office did not use as a precedent for permitting other media outlets to also do so. The government made a general announcement on radar in April 1943, and government and military officials frequently leaked information on the subject, but restrictions did not end until the day after Japan's surrender in August 1945.[1]:195–197
…
After Hiroshima The New York Times was first to report on the specifics of the Manhattan Project on August 7, 1945, saying the bomb was built in "three 'hidden cities' with a total population of 100,000 inhabitants"; Los Alamos, Oak Ridge, Tennessee, and Hanford, Washington. "None of the people, who came to these developments from homes all the way from Maine to California, had the slightest idea of what they were making in the gigantic Government plants they saw around them," the New York Times said.
https://en.wikipedia.org/wiki/Nuclear_reactor
Retrieved: 12 September 2017
Archive: https://archive.is/UU4vE
Nuclear Reactor
A nuclear reactor, formerly known as an atomic pile, is a device used to initiate and control a sustained nuclear chain reaction. Nuclear reactors are used at nuclear power plants for electricity generation and in propulsion of ships. Heat from nuclear fission is passed to a working fluid (water or gas), which runs through steam turbines. These either drive a ship's propellers or turn electrical generators. Nuclear generated steam in principle can be used for industrial process heat or for district heating. Some reactors are used to produce isotopes for medical and industrial use, or for production of weapons-grade plutonium. Some are run only for research. As of April 2014, the IAEA reports there are 435 nuclear power reactors in operation, in 31 countries around the world.[1]
Core of CROCUS, a small nuclear reactor used for research at the EPFL in Switzerland
…
An induced nuclear fission event. A neutron is absorbed by the nucleus of a uranium-235 atom, which in turn splits into fast-moving lighter elements (fission products) and free neutrons. Though both reactors and nuclear weapons rely on nuclear chain-reactions, the rate of reactions in a reactor occurs much more slowly than in a bomb.
…
The Chicago Pile, the first nuclear reactor, built in secrecy at the University of Chicago in 1942 during World War II as part of the US's Manhattan project.
…
Classification by type of nuclear reaction[edit]
Nuclear fission[edit]
All commercial power reactors are based on nuclear fission. They generally use uranium and its product plutonium as nuclear fuel, though a thorium fuel cycle is also possible. Fission reactors can be divided roughly into two classes, depending on the energy of the neutrons that sustain the fission chain reaction:
• Thermal reactors (the most common type of nuclear reactor) use slowed or thermal neutrons to keep up the fission of their fuel. Almost all current reactors are of this type. These contain neutron moderator materials that slow neutrons until their neutron temperature is thermalized, that is, until their kinetic energy approaches the average kinetic energy of the surrounding particles.
…
• Fast neutron reactors use fast neutrons to cause fission in their fuel.
…
Nuclear fusion[edit]
Fusion power is an experimental technology, generally with hydrogen as fuel. While not suitable for power production, Farnsworth-Hirsch fusors are used to produce neutron radiation.
Classification by moderator material[edit]
Used by thermal reactors:
• Graphite-moderated reactors
• Water moderated reactors
• Heavy-water reactors (Used in Canada, India, Argentina, China, Pakistan, Romania and South Korea).[21])
• Light-water-moderated reactors (LWRs). Light-water reactors (the most common type of thermal reactor) use ordinary water to moderate and cool the reactors. When at operating temperature, if the temperature of the water increases, its density drops, and fewer neutrons passing through it are slowed enough to trigger further reactions. That negative feedback stabilizes the reaction rate. Graphite and heavy-water reactors tend to be more thoroughly thermalized than light water reactors. Due to the extra thermalization, these types can use natural uranium/unenriched fuel.
• Light-element-moderated reactors.
• Molten salt reactors (MSRs) are moderated by light elements such as lithium or beryllium, which are constituents of the coolant/fuel matrix salts LiF and BeF2.
• Liquid metal cooled reactors, such as those whose coolant is a mixture of lead and bismuth, may use BeO as a moderator.
• Organically moderated reactors (OMR) use biphenyl and terphenyl as moderator and coolant.…
In thermal nuclear reactors (LWRs in specific), the coolant acts as a moderator that must slow down the neutrons before they can be efficiently absorbed by the fuel.
…
Nuclear safety concerns and controversy[edit]
Main article: Nuclear safetyNuclear safety covers the actions taken to prevent nuclear and radiation accidents and incidents or to limit their consequences. The nuclear power industry has improved the safety and performance of reactors, and has proposed new safer (but generally untested) reactor designs but there is no guarantee that the reactors will be designed, built and operated correctly.[34] Mistakes do occur and the designers of reactors at Fukushima in Japan did not anticipate that a tsunami generated by an earthquake would disable the backup systems that were supposed to stabilize the reactor after the earthquake,[35] despite multiple warnings by the NRG and the Japanese nuclear safety administration.[citation needed] According to UBS AG, the Fukushima I nuclear accidents have cast doubt on whether even an advanced economy like Japan can master nuclear safety.[36] Catastrophic scenarios involving terrorist attacks are also conceivable.[34] An interdisciplinary team from MIT has estimated that given the expected growth of nuclear power from 2005–2055, at least four serious nuclear accidents would be expected in that period.[37]
MES Note: Given the terrorist attack on 9/11 by the USA and Co., don't be surprised if some nuclear type "accidents" or "terrorist attacks" occur in the near future…
Also note the line about even an "advanced economy" like Japan can't ensure nuclear safety… This again ties with the theme of 9/11 and suppression of game-changing energy sources!
Nuclear accidents and controversy[edit]
See also: Lists of nuclear disasters and radioactive incidentsSome serious nuclear and radiation accidents have occurred. Nuclear power plant accidents include the SL-1 accident (1961), the Three Mile Island accident (1979), Chernobyl disaster (1986), and the Fukushima Daiichi nuclear disaster (2011).[39]Nuclear-powered submarine mishaps include the K-19 reactor accident (1961),[40] the K-27 reactor accident (1968),[41] and the K-431 reactor accident (1985).[39]
Nuclear reactors have been launched into Earth orbit at least 34 times. A number of incidents connected with the unmanned nuclear-reactor-powered Soviet RORSAT radar satellite program resulted in spent nuclear fuel re-entering the Earth's atmosphere from orbit.[citation needed]
Three of the reactors at Fukushima I overheated, causing meltdowns that eventually led to explosions, which released large amounts of radioactive material into the air.[38]
https://en.wikipedia.org/wiki/Fukushima_Daiichi_nuclear_disaster
Retrieved: 12 September 2017
Archive: https://archive.is/dNG2F
Fukushima Daiichi nuclear disaster
The Fukushima Daiichi nuclear disaster (???????????? Fukushima Dai-ichi (pronunciation) genshiryoku hatsudensho jiko) was an energy accident at the Fukushima Daiichi Nuclear Power Plant in Fukushima, initiated primarily by the tsunami following the Tohoku earthquake on 11 March 2011.[6] Immediately after the earthquake, the active reactors automatically shut down their sustained fission reactions. However, the tsunami disabled the emergency generators that would have provided power to control and operate the pumps necessary to cool the reactors. The insufficient cooling led to three nuclear meltdowns, hydrogen-air explosions, and the release of radioactive material in Units 1, 2, and 3 from 12 March to 15 March. Loss of cooling also caused the pool for storing spent fuel from Reactor 4 to overheat on 15 March due to the decay heat from the fuel rods.
On 5 July 2012, the Fukushima Nuclear Accident Independent Investigation Commission (NAIIC) found that the causes of the accident had been foreseeable, and that the plant operator, Tokyo Electric Power Company (TEPCO), had failed to meet basic safety requirements such as risk assessment, preparing for containing collateral damage, and developing evacuation plans. On 12 October 2012, TEPCO admitted for the first time that it had failed to take necessary measures for fear of inviting lawsuits or protests against its nuclear plants.[7][8][9][10]
MES Note: This whole narrative sounds fishy… And the coverup of 9/11 suggests that by default we must be always skeptical of any "official" mainstream narrative.
The Fukushima disaster was the most significant nuclear incident since April 26, 1986 the Chernobyl disaster and the second disaster to be given the Level 7 event classification of the International Nuclear Event Scale.[11] Though there have been no fatalities linked to radiation due to the accident, the eventual number of cancer deaths, according to the linear no-threshold theory of radiation safety, that will be caused by the accident is expected to be around 130–640 people in the years and decades ahead.[12][13][14] The United Nations Scientific Committee on the Effects of Atomic Radiation[15]and World Health Organization report that there will be no increase in miscarriages, stillbirths or physical and mental disorders in babies born after the accident.[16] However, an estimated 1,600 deaths are believed to have occurred due to the resultant evacuation conditions.[17][18] There are no clear plans for decommissioning the plant, but the plant management estimate is 30 or 40 years.[19]
MES Note: I don't trust the United Nation speculation or statistics for just one second…
…
…
Map of contaminated areas around the plant (22 March – 3 April 2011)
Now let's finally get to main topic I wanted to cover, Cold Fusion!
I alluded to Cold Fusion in Part 1 as likely being a suppressed Free Energy technology, but now let's take a closer look!
https://en.wikipedia.org/wiki/Cold_fusion
Retrieved: 8 September 2017
Archive: https://archive.is/at1KM
Cold Fusion
Cold fusion is a hypothesized type of nuclear reaction that would occur at, or near, room temperature. This is compared with the "hot" fusion which takes place naturally within stars, under immense pressure and at temperatures of millions of degrees, and distinguished from muon-catalyzed fusion. There is currently no accepted theoretical model that would allow cold fusion to occur.
In 1989 Martin Fleischmann (then one of the world's leading electrochemists) and Stanley Pons reported that their apparatus had produced anomalous heat ("excess heat") of a magnitude they asserted would defy explanation except in terms of nuclear processes.[1] They further reported measuring small amounts of nuclear reaction byproducts, including neutrons and tritium.[2] The small tabletop experiment involved electrolysis of heavy water on the surface of a palladium (Pd) electrode.[3] The reported results received wide media attention,[3] and raised hopes of a cheap and abundant source of energy.[4]
Many scientists tried to replicate the experiment with the few details available. Hopes faded due to the large number of negative replications, the withdrawal of many reported positive replications, the discovery of flaws and sources of experimental error in the original experiment, and finally the discovery that Fleischmann and Pons had not actually detected nuclear reaction byproducts.[5] By late 1989, most scientists considered cold fusion claims dead,[6][7] and cold fusion subsequently gained a reputation as pathological science.[8][9] In 1989 the United States Department of Energy (DOE) concluded that the reported results of excess heat did not present convincing evidence of a useful source of energy and decided against allocating funding specifically for cold fusion. A second DOE review in 2004, which looked at new research, reached similar conclusions and did not result in DOE funding of cold fusion.[10]
MES Note: Wow… Given that the government has "allocated" trillions to unending wars and staging fabricated terror events such as 9/11, topics they avoid (publicly) funding are areas we definitely need to investigate further!
A small community of researchers continues to investigate cold fusion,[6][11] now often preferring the designation low-energy nuclear reactions (LENR) or condensed matter nuclear science (CMNS).[12][13][14][15] Since cold fusion articles are rarely published in peer-reviewed mainstream scientific journals, they do not attract the level of scrutiny expected for mainstream scientific publications.[16]
MES Note: Yours truly is part of these researchers.
Also, 9/11 proves that these mainstream "scientific" journals are complete jokes!
History
Nuclear fusion is normally understood to occur at temperatures in the tens of millions of degrees. Since the 1920s, there has been speculation that nuclear fusion might be possible at much lower temperatures by catalytically fusing hydrogen absorbed in a metal catalyst. In 1989, a claim by Stanley Pons and Martin Fleischmann (then one of the world's leading electrochemists) that such cold fusion had been observed…
…
Early research
The ability of palladium to absorb hydrogen was recognized as early as the nineteenth century by Thomas Graham.[17][18] In the late 1920s, two Austrian born scientists, Friedrich Paneth and Kurt Peters, originally reported the transformation of hydrogen into helium by nuclear catalysis when hydrogen was absorbed by finely divided palladium at room temperature. However, the authors later retracted that report, saying that the helium they measured was due to background from the air.[17][19]
In 1927 Swedish scientist John Tandberg reported that he had fused hydrogen into helium in an electrolytic cell with palladium electrodes.[17] On the basis of his work, he applied for a Swedish patent for "a method to produce helium and useful reaction energy".[17] Due to Paneth and Peters's retraction and his inability to explain the physical process, his patent application was denied.[17][20] After deuterium was discovered in 1932, Tandberg continued his experiments with heavy water.[17] The final experiments made by Tandberg with heavy water were similar to the original experiment by Fleischmann and Pons.[21] Fleischmann and Pons were not aware of Tandberg's work.[22][text 1][text 2]
The term "cold fusion" was used as early as 1956 in a New York Times article about Luis Alvarez's work on muon-catalyzed fusion.[23] Paul Palmer and then Steven Jones of Brigham Young University used the term "cold fusion" in 1986 in an investigation of "geo-fusion", the possible existence of fusion involving hydrogen isotopes in a planetary core.[24]
…
Fleischmann–Pons experiment
The most famous cold fusion claims were made by Stanley Pons and Martin Fleischmann in 1989. After a brief period of interest by the wider scientific community, their reports were called into question by nuclear physicists. Pons and Fleischmann never retracted their claims, but moved their research program to France after the controversy erupted.
Events preceding announcement
Martin Fleischmann of the University of Southampton and Stanley Pons of the University of Utah hypothesized that the high compression ratio and mobility of deuterium that could be achieved within palladium metal using electrolysis might result in nuclear fusion.[26] To investigate, they conducted electrolysis experiments using a palladium cathode and heavy water within a calorimeter, an insulated vessel designed to measure process heat. Current was applied continuously for many weeks, with the heavy water being renewed at intervals.[26] Some deuterium was thought to be accumulating within the cathode, but most was allowed to bubble out of the cell, joining oxygen produced at the anode.[27] For most of the time, the power input to the cell was equal to the calculated power leaving the cell within measurement accuracy, and the cell temperature was stable at around 30 °C. But then, at some point (in some of the experiments), the temperature rose suddenly to about 50 °C without changes in the input power. These high temperature phases would last for two days or more and would repeat several times in any given experiment once they had occurred. The calculated power leaving the cell was significantly higher than the input power during these high temperature phases. Eventually the high temperature phases would no longer occur within a particular cell.[27]
In 1988 Fleischmann and Pons applied to the United States Department of Energy for funding towards a larger series of experiments. Up to this point they had been funding their experiments using a small device built with $100,000 out-of-pocket.[28] The grant proposal was turned over for peer review, and one of the reviewers was Steven Jones of Brigham Young University.[28] Jones had worked for some time on muon-catalyzed fusion, a known method of inducing nuclear fusion without high temperatures, and had written an article on the topic entitled "Cold nuclear fusion" that had been published in Scientific American in July 1987. Fleischmann and Pons and co-workers met with Jones and co-workers on occasion in Utah to share research and techniques. During this time, Fleischmann and Pons described their experiments as generating considerable "excess energy", in the sense that it could not be explained by chemical reactions alone.[27] They felt that such a discovery could bear significant commercial value and would be entitled to patent protection. Jones, however, was measuring neutron flux, which was not of commercial interest.[28][clarification needed] To avoid future problems, the teams appeared to agree to simultaneously publish their results, though their accounts of their 6 March meeting differ.[29]
MES Note: Lifelong government disinfo and controlled opposition agent, whom was also behind the bogus "thermite" theory to coverup the #FreeEnergy tech used on 9/11, Steven Jones just so happens to be one of the grant reviewers of Pons and Flesichmann's Cold Fusion work!
Not only that but Jones was receiving grant money for the same field that Pons and Fleishmann were in, and thus reviewing the grant gave him access to all of their data, hence the issues with timing of their publishing!!
Steven Jones also is a founding member of the controlled opposition Architects & Engineers for 9/11 Truth, which is an absolute disgrace of an organization.
Follow my # 911Truth Video Series as I will be covering Jones and AE 9/11 "Truth"! https://mes.fm/911truth-playlist.
Electrolysis cell schematic
Announcement
In mid-March 1989, both research teams were ready to publish their findings, and Fleischmann and Jones had agreed to meet at an airport on 24 March to send their papers to Nature via FedEx.[29] Fleischmann and Pons, however, pressured by the University of Utah, which wanted to establish priority on the discovery,[30] broke their apparent agreement, submitting their paper to the Journal of Electroanalytical Chemistry on 11 March, and disclosing their work via a press release[31] and press conference on 23 March.[28] Jones, upset, faxed in his paper to Nature after the press conference.[29]
MES Note: Notice how the pathetic government controlled Wikipedia make it appear as if Jones is the victim when in fact Jones should be thrown underneath a prison.
To learn more about how Steven Jones helped sabotaged Pons and Fleishmann's work, as well as diverting attention from 9/11 #FreeEnergy, and his closes ties to the Department of Energy and 1980s' research at Los Alamos Laboratory, go to this webpage!
http://drjudywood.com/articles/JJ/JJ7.html
Retrieved: 15 September 2017
Archive: https://archive.is/OWt75
Dr. Eugene Mallove and Dr. Judy Wood's student Michael Zebuhr were murdered because they were dangerous to the establishment.
To learn even more about the whole Cold Fusion almost certain cover-up, make sure to watch the following documentary!
Retrieved: 15 September 2017
Archive: https://archive.is/B4hZd
Note: For a version with subtitles although video quality is not as good:
# ILikeSubtitles
From media smears and lies, to outright falsification of data to "debunk" cold fusion, the establishment will go to great lengths to keep people enslaved! # WakeUp
Former MIT Engineer Eugene Mallove resigned in protest against MIT falsifying data when trying to reproduce cold fusion… More on Mallove later in this video…
Also, Dr. Eugene Mallove helped write the documentary!
http://www.imdb.com/title/tt1217017/
Retrieved: 15 September 2017
Archive: https://archive.is/5x9bz
Wikipedia page continued:
Fleischmann and Pons' announcement drew wide media attention.[32] But the 1986 discovery of high-temperature superconductivity had made the scientific community more open to revelations of unexpected scientific results that could have huge economic repercussions and that could be replicated reliably even if they had not been predicted by established theories.[33] Many scientists were also reminded of the Mössbauer effect, a process involving nuclear transitions in a solid. Its discovery 30 years earlier had also been unexpected, though it was quickly replicated and explained within the existing physics framework.[34]
The announcement of a new purported clean source of energy came at a crucial time: adults still remembered the 1973 oil crisis and the problems caused by oil dependence, anthropogenic global warming was starting to become notorious, the anti-nuclear movement was labeling nuclear power plants as dangerous and getting them closed, people had in mind the consequences of strip mining, acid rain, the greenhouse effect and the Exxon Valdez oil spill, which happened the day after the announcement.[35] In the press conference, Chase N. Peterson, Fleischmann and Pons, backed by the solidity of their scientific credentials, repeatedly assured the journalists that cold fusion would solve environmental problems, and would provide a limitless inexhaustible source of clean energy, using only seawater as fuel.[36] They said the results had been confirmed dozens of times and they had no doubts about them.[37] In the accompanying press release Fleischmann was quoted saying: "What we have done is to open the door of a new research area, our indications are that the discovery will be relatively easy to make into a usable technology for generating heat and power, but continued work is needed, first, to further understand the science and secondly, to determine its value to energy economics."[31]
Response and fallout
Although the experimental protocol had not been published, physicists in several countries attempted, and failed, to replicate the excess heat phenomenon. The first paper submitted to Nature reproducing excess heat, although it passed peer-review, was rejected because most similar experiments were negative and there were no theories that could explain a positive result;[notes 1][38] this paper was later accepted for publication by the journal Fusion Technology. Nathan Lewis, professor of chemistry at the California Institute of Technology, led one of the most ambitious validation efforts, trying many variations on the experiment without success,[39] while CERN physicist Douglas R. O. Morrison said that "essentially all" attempts in Western Europe had failed.[6] Even those reporting success had difficulty reproducing Fleischmann and Pons' results.[40] On 10 April 1989, a group at Texas A&M University published results of excess heat and later that day a group at the Georgia Institute of Technology announced neutron production—the strongest replication announced up to that point due to the detection of neutrons and the reputation of the lab.[41] On 12 April Pons was acclaimed at an ACS meeting.[41] But Georgia Tech retracted their announcement on 13 April, explaining that their neutron detectors gave false positives when exposed to heat.[42] Another attempt at independent replication, headed by Robert Huggins at Stanford University, which also reported early success with a light water control,[43] became the only scientific support for cold fusion in 26 April US Congress hearings.[text 3] But when he finally presented his results he reported an excess heat of only one degree celsius, a result that could be explained by chemical differences between heavy and light water in the presence of lithium.[notes 2] He had not tried to measure any radiation[44] and his research was derided by scientists who saw it later.[45] For the next six weeks, competing claims, counterclaims, and suggested explanations kept what was referred to as "cold fusion" or "fusion confusion" in the news.[29][46]
In April 1989, Fleischmann and Pons published a "preliminary note" in the Journal of Electroanalytical Chemistry.[26] This paper notably showed a gamma peak without its corresponding Compton edge, which indicated they had made a mistake in claiming evidence of fusion byproducts.[47] Fleischmann and Pons replied to this critique,[48] but the only thing left clear was that no gamma ray had been registered and that Fleischmann refused to recognize any mistakes in the data.[49] A much longer paper published a year later went into details of calorimetry but did not include any nuclear measurements.[27]
Nevertheless, Fleischmann and Pons and a number of other researchers who found positive results remained convinced of their findings.[6] The University of Utah asked Congress to provide $25 million to pursue the research, and Pons was scheduled to meet with representatives of President Bush in early May.[6]
On 30 April 1989 cold fusion was declared dead by the New York Times. The Times called it a circus the same day, and the Boston Herald attacked cold fusion the following day.[50]
On 1 May 1989 the American Physical Society held a session on cold fusion in Baltimore, including many reports of experiments that failed to produce evidence of cold fusion. At the end of the session, eight of the nine leading speakers stated that they considered the initial Fleischmann and Pons claim dead, with the ninth, Johann Rafelski, abstaining.[6] Steven E. Koonin of Caltech called the Utah report a result of "the incompetence and delusion of Pons and Fleischmann," which was met with a standing ovation.[51] Douglas R. O. Morrison, a physicist representing CERN, was the first to call the episode an example of pathological science.[6][52]
On 4 May, due to all this new criticism, the meetings with various representatives from Washington were cancelled.[53]
From 8 May only the A&M tritium results kept cold fusion afloat.[54]
In July and November 1989, Nature published papers critical of cold fusion claims.[55][56] Negative results were also published in several other scientific journals including Science, Physical Review Letters, and Physical Review C (nuclear physics).[notes 3]
In August 1989, in spite of this trend, the state of Utah invested $4.5 million to create the National Cold Fusion Institute.[57]
The United States Department of Energy organized a special panel to review cold fusion theory and research.[58] The panel issued its report in November 1989, concluding that results as of that date did not present convincing evidence that useful sources of energy would result from the phenomena attributed to cold fusion.[59] The panel noted the large number of failures to replicate excess heat and the greater inconsistency of reports of nuclear reaction byproducts expected by established conjecture. Nuclear fusion of the type postulated would be inconsistent with current understanding and, if verified, would require established conjecture, perhaps even theory itself, to be extended in an unexpected way. The panel was against special funding for cold fusion research, but supported modest funding of "focused experiments within the general funding system."[60] Cold fusion supporters continued to argue that the evidence for excess heat was strong, and in September 1990 the National Cold Fusion Institute listed 92 groups of researchers from 10 different countries that had reported corroborating evidence of excess heat, but they refused to provide any evidence of their own arguing that it could endanger their patents.[61]However, no further DOE nor NSF funding resulted from the panel's recommendation.[62] By this point, however, academic consensus had moved decidedly toward labeling cold fusion as a kind of "pathological science".[8][63]
In March 1990 Dr. Michael H. Salamon, a physicist from the University of Utah, and nine co-authors reported negative results.[64] University faculty were then "stunned" when a lawyer representing Pons and Fleischmann demanded the Salamon paper be retracted under threat of a lawsuit. The lawyer later apologized; Fleischmann defended the threat as a legitimate reaction to alleged bias displayed by cold-fusion critics.[65]
In early May 1990 one of the two A&M researchers, Kevin Wolf, acknowledged the possibility of spiking, but said that the most likely explanation was tritium contamination in the palladium electrodes or simply contamination due to sloppy work.[66] In June 1990 an article in Science by science writer Gary Taubes destroyed the public credibility of the A&M tritium results when it accused its group leader John Bockris and one of his graduate students of spiking the cells with tritium.[67] In October 1990 Wolf finally said that the results were explained by tritium contamination in the rods.[68] An A&M cold fusion review panel found that the tritium evidence was not convincing and that, while they couldn't rule out spiking, contamination and measurements problems were more likely explanations,[text 4] and Bockris never got support from his faculty to resume his research.
On 30 June 1991 the National Cold Fusion Institute closed after it ran out of funds;[69] it found no excess heat, and its reports of tritium production were met with indifference.[70]
On 1 January 1991 Pons left the University of Utah and went to Europe.[70][71] In 1992, Pons and Fleischman resumed research with Toyota Motor Corporation's IMRA lab in France.[70] Fleischmann left for England in 1995, and the contract with Pons was not renewed in 1998 after spending $40 million with no tangible results.[72] The IMRA laboratory stopped cold fusion research in 1998 after spending £12 million.[3] Pons has made no public declarations since, and only Fleischmann continued giving talks and publishing papers.[72]
MES Note: This entire narrative seems very fishy as I could not find the actual sources to these figures.
Also why would IMRA stop research funding after $40 million (or £12 million?) with no "tangible" results…… why not stop after $20 million? After $10 million? After $1 million??
Remember that Pons & Fleischmann's initial work involved $100,000 out of pocket, so it seems HIGHLY unlikely that IMRA would sign a large multi-million dollar contract… What is this professional sports??!! # DontYouBelieveIt
Also, this narrative is in stark contrast to the words of Martin Fleischmann himself from the above documentary:
Note: The screenshot is from the video link that had subtitles.
My view is that the Wikipedia narrative is just another typical establishment distortion of reality, and that Fleishmann was almost certainly stopped before he was able to make a commercial device!
Also, the Wikipedia page cites the 1993 book "Bad Science: The Short Life and Weird Times of Cold Fusion" by Gary Taubes in presenting this awful distorted narrative, and even for some of the 1998 dates… ??
It is my opinion that Gary Taubes is just another government/CIA asset used to give "independent" reviews of topics they want buried or discredited; and then the government controlled Wikipedia "cites" that book instead of the ACTUAL source of any point they want to reference… # UNBELIEVABLE
Mostly in the 1990s, several books were published that were critical of cold fusion research methods and the conduct of cold fusion researchers.[73] Over the years, several books have appeared that defended them.[74] Around 1998, the University of Utah had already dropped its research after spending over $1 million, and in the summer of 1997, Japan cut off research and closed its own lab after spending $20 million.[75]
…
Current research
Cold fusion research continues today in a few specific venues, but the wider scientific community has generally marginalized the research being done and researchers have had difficulty publishing in mainstream journals.[6][7][11] The remaining researchers often term their field Low Energy Nuclear Reactions (LENR), Chemically Assisted Nuclear Reactions (CANR),[79] Lattice Assisted Nuclear Reactions (LANR), Condensed Matter Nuclear Science (CMNS) or Lattice Enabled Nuclear Reactions; one of the reasons being to avoid the negative connotations associated with "cold fusion".[77][80] The new names avoid making bold implications, like implying that fusion is actually occurring.[81]
MES Note: Here at MES, I like the term Cold Fusion because it is a badge of honor to be "ridiculed" by the pathetic mainstream establishment.
The researchers who continue acknowledge that the flaws in the original announcement are the main cause of the subject's marginalization, and they complain of a chronic lack of funding[82] and no possibilities of getting their work published in the highest impact journals.[83] University researchers are often unwilling to investigate cold fusion because they would be ridiculed by their colleagues and their professional careers would be at risk.[84]
MES Note: Nothing says "science" like being afraid for your lively hood to simply research into hidden technology while criminals use #FreeEnergy tech to murder thousands in order to justify killing millions more… # 911Truth # WakeUpPeople!
…
One of the main criticisms of cold fusion was that deuteron-deuteron fusion into helium was expected to result in the production of gamma rays—which were not observed and were not observed in subsequent cold fusion experiments.[40][123] Cold fusion researchers have since claimed to find X-rays, helium, neutrons[124] and nuclear transmutations.[125]
…
Reproducibility
In 1989, after Fleischmann and Pons had made their claims, many research groups tried to reproduce the Fleischmann-Pons experiment, without success. A few other research groups, however, reported successful reproductions of cold fusion during this time. In July 1989, an Indian group from the Bhabha Atomic Research Centre (P. K. Iyengar and M. Srinivasan) and in October 1989, John Bockris' group from Texas A&M University reported on the creation of tritium. In December 1990, professor Richard Oriani of the University of Minnesota reported excess heat.[142]
Groups that did report successes found that some of their cells were producing the effect, while other cells that were built exactly the same and used the same materials were not producing the effect.[143] Researchers that continued to work on the topic have claimed that over the years many successful replications have been made, but still have problems getting reliable replications.[144] Reproducibility is one of the main principles of the scientific method, and its lack led most physicists to believe that the few positive reports could be attributed to experimental error.[143][text 9]
…
Publications
The ISI identified cold fusion as the scientific topic with the largest number of published papers in 1989, of all scientific disciplines.[158] The Nobel Laureate Julian Schwinger declared himself a supporter of cold fusion in the fall of 1989, after much of the response to the initial reports had turned negative. He tried to publish his theoretical paper "Cold Fusion: A Hypothesis" in Physical Review Letters, but the peer reviewers rejected it so harshly that he felt deeply insulted, and he resigned from the American Physical Society (publisher of PRL) in protest.[159]
The number of papers sharply declined after 1990 because of two simultaneous phenomena:[158] scientists abandoning the field and journal editors declining to review new papers, and cold fusion fell off the ISI charts.[158][160] Researchers who got negative results abandoned the field, while others kept publishing.[161] A 1993 paper in Physics Letters A was the last paper published by Fleischmann, and "one of the last reports [by Fleischmann] to be formally challenged on technical grounds by a cold fusion skeptic".[text 10]
The Journal of Fusion Technology (FT) established a permanent feature in 1990 for cold fusion papers, publishing over a dozen papers per year and giving a mainstream outlet for cold fusion researchers. When editor-in-chief George H. Miley retired in 2001, the journal stopped accepting new cold fusion papers.[160] This has been cited as an example of the importance of sympathetic influential individuals to the publication of cold fusion papers in certain journals.[160]
The decline of publications in cold fusion has been described as a "failed information epidemic".[text 11] The sudden surge of supporters until roughly 50% of scientists support the theory, followed by a decline until there is only a very small number of supporters, has been described as a characteristic of pathological science.[text 12][notes 4]
…
Conferences
Cold fusion researchers were for many years unable to get papers accepted at scientific meetings, prompting the creation of their own conferences. The first International Conference on Cold Fusion (ICCF) was held in 1990, and has met every 12 to 18 months since. Attendees at some of the early conferences were described as offering no criticism to papers and presentations for fear of giving ammunition to external critics;[164] thus allowing the proliferation of crackpots and hampering the conduct of serious science.[165] Critics and skeptics stopped attending these conferences, with the notable exception of Douglas Morrison,[166] who died in 2001.
Patents
Although details have not surfaced, it appears that the University of Utah forced the 23 March 1989 Fleischmann and Pons announcement to establish priority over the discovery and its patents before the joint publication with Jones.[30] The Massachusetts Institute of Technology (MIT) announced on 12 April 1989 that it had applied for its own patents based on theoretical work of one of its researchers, Peter L. Hagelstein, who had been sending papers to journals from the 5 to 12 April.[174] On 2 December 1993 the University of Utah licensed all its cold fusion patents to ENECO, a new company created to profit from cold fusion discoveries,[175] and in March 1998 it said that it would no longer defend its patents.[75]
The U.S. Patent and Trademark Office (USPTO) now rejects patents claiming cold fusion.[88] Esther Kepplinger, the deputy commissioner of patents in 2004, said that this was done using the same argument as with perpetual motion machines: that they do not work.[88]
MES Note: Cold Fusion and Perpetual Motion Machines can't be patented… Wow!
I rather not give the government knowledge of advanced tech anyways…
Patent applications are required to show that the invention is "useful", and this utility is dependent on the invention's ability to function.[176] In general USPTO rejections on the sole grounds of the invention's being "inoperative" are rare, since such rejections need to demonstrate "proof of total incapacity",[176] and cases where those rejections are upheld in a Federal Court are even rarer: nevertheless, in 2000, a rejection of a cold fusion patent was appealed in a Federal Court and it was upheld, in part on the grounds that the inventor was unable to establish the utility of the invention.[176][notes 5]
MES Note: UNBELIEVABLE! How can a cold fusion device EVER be deemed "useless"… # WOW
A U.S. patent might still be granted when given a different name to disassociate it from cold fusion,[177] though this strategy has had little success in the US: the same claims that need to be patented can identify it with cold fusion, and most of these patents cannot avoid mentioning Fleischmann and Pons' research due to legal constraints, thus alerting the patent reviewer that it is a cold-fusion-related patent.[177] David Voss said in 1999 that some patents that closely resemble cold fusion processes, and that use materials used in cold fusion, have been granted by the USPTO.[178] The inventor of three such patents had his applications initially rejected when they were reviewed by experts in nuclear science; but then he rewrote the patents to focus more in the electrochemical parts so they would be reviewed instead by experts in electrochemistry, who approved them.[178][179] When asked about the resemblance to cold fusion, the patent holder said that it used nuclear processes involving "new nuclear physics" unrelated to cold fusion.[178] Melvin Miles was granted in 2004 a patent for a cold fusion device, and in 2007 he described his efforts to remove all instances of "cold fusion" from the patent description to avoid having it rejected outright.[180]
At least one patent related to cold fusion has been granted by the European Patent Office.[181]
A patent only legally prevents others from using or benefiting from one's invention. However, the general public perceives a patent as a stamp of approval, and a holder of three cold fusion patents said the patents were very valuable and had helped in getting investments.[178]
Cultural references
In Undead Science, sociologist Bart Simon gives some examples of cold fusion in popular culture, saying that some scientists use cold fusion as a synonym for outrageous claims made with no supporting proof,[182] and courses of ethics in science give it as an example of pathological science.[182] It has appeared as a joke in Murphy Brown and The Simpsons.[182] It was adopted as a software product name Adobe Cold Fusion and a brand of protein bars (Cold Fusion Foods).[182] It has also appeared in advertising as a synonym for impossible science, for example a 1995 advertisement for Pepsi Max.[182]
https://en.wikipedia.org/wiki/Electrolysis
Retrieved: 15 September 2017
Archive: https://archive.is/MqiMz
Electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses a direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential.
Illustration of an electrolysis apparatus used in a school laboratory.
https://en.wikipedia.org/wiki/Martin_Fleischmann
Retrieved: 12 September 2017
Archive: https://archive.is/acn7E
Martin Fleischmann
Martin Fleischmann FRS (29 March 1927 – 3 August 2012) was a British chemist noted for his work in electrochemistry.[3][4] Premature announcement of his cold fusion research with Stanley Pons,[5] regarding excess heat in heavy water, caused a media sensation although they continued their interest and research in cold fusion.[6][citation needed]
Fleischmann showing part of his cold fusion test apparatus
…
Retirement (1992 – 2012)[edit]
In 1992, Fleischmann moved to France with Pons to continue their work at the IMRA laboratory (part of Technova Corporation, a subsidiary of Toyota), but in 1995 he retired and returned to England.[32][33] He co-authored further papers with researchers from the US Navy[34][35] and Italian national laboratories (INFN and ENEA),[36]on the subject of cold fusion.
…
Death[edit]
Fleischmann died at home in Tisbury, Wiltshire on 3 August 2012, of natural causes. He had suffered from Parkinson's disease, diabetes and heart disease.[4]
…
Legacy[edit]
While holding the Faraday Chair of Electrochemistry he and Graham Hills established in the late 60s the now renowned Electrochemistry Group of the University of Southampton.[7]
Fleischmann produced over 272 scientific papers and book chapters on the field of electrochemistry.[7]
…
Peer-reviewed papers on "Cold Fusion"
• Fleischmann, Martin; Pons, Stanley; Anderson, Mark W.; Li, Lian Jun; Hawkins, Marvin (1990). "Calorimetry of the palladium-deuterium-heavy water system". Journal of Electroanalytical Chemistry. 287 (2): 293–348. doi:10.1016/0022-0728(90)80009-U.
• Fleischmann, Martin; Pons, Stanley (1992). "Some Comments on The Paper 'Analysis of Experiments on The Calorimetry of LiOD-D2O Electrochemical Cells,' R.H. Wilson et al., Journal of Electroanalytical Chemistry, Vol. 332, (1992)". Journal of Electroanalytical Chemistry. 332: 33–53. doi:10.1016/0022-0728(92)80339-6.
• Fleischmann, Martin; Pons, S (1993). "Calorimetry of the Pd-D2O system: from simplicity via complications to simplicity". Physics Letters A. 176 (1–2): 118–129. Bibcode:1993PhLA..176..118F. doi:10.1016/0375-9601(93)90327-V.
https://en.wikipedia.org/wiki/Stanley_Pons
Retrieved: 12 September 2017
Archive: https://archive.is/tUe0i
Stanley Pons
Bobby Stanley Pons (born August 23, 1943) is an American-born French electrochemist known for his work with Martin Fleischmann on cold fusion in the 1980s and 1990s.[3]
…
Career
On March 23, 1989, while Pons was the chairman of the chemistry department at the University of Utah,[4] he and Fleischmann announced the experimental production of "N-Fusion", which was quickly labeled by the press as cold fusion.[5]After a short period of public acclaim, hundreds of scientists attempted to reproduce the effects but generally failed.[6] After the claims were found to be unreproducible, the scientific community determined the claims were incomplete and inaccurate.[1][6][7][7][8][9][10]
Pons moved to France in 1992, along with Fleischmann, to work at a Toyota-sponsored laboratory. The laboratory closed in 1998 after a £12 million research investment without conclusive results.[2] He gave up his US citizenship[11] and became a French citizen.[12]
https://en.wikipedia.org/wiki/Eugene_Mallove
Retrieved: 9 August 2017
Archive: https://archive.is/dSQbx
Eugene Mallove
Eugene Franklin Mallove (June 9, 1947 – May 14, 2004) was an American scientist, science writer, editor, and publisher of Infinite Energy magazine, and founder of the non-profit organization New Energy Foundation. He was a proponent of cold fusion, and a supporter of its research and related exploratory alternative energy topics, several of which are sometimes characterised as "fringe science".
Mallove authored Fire from Ice, a book detailing the 1989 report of table-top cold fusion from Stanley Pons and Martin Fleischmann at the University of Utah. Among other things, the book advances a conspiracy theory, claiming that the team did produce "greater-than-unity" output energy in an experiment successfully replicated on several occasions, but that the results were suppressed through an organized campaign of ridicule from mainstream physicists, including those studying controlled thermonuclear fusion, trying to protect their research and funding.
Biography
Eugene Mallove held a BS (1969) and MS degree (1970) in aeronautical and astronautical engineering from MIT and a ScD degree (1975) in environmental health sciences from Harvard University. He had worked for technology engineering firms such as Hughes Research Laboratories, the Analytic Science Corporation, and MIT's Lincoln Laboratory, and he consulted in research and development of new energies.
…
Mallove was murdered in 2004 while cleaning out his former childhood home, which had been rented out. Three people have been arrested and charged in connection with the killing; two were convicted of first-degree manslaughter[1] and murder,[2] in 2012 and 2014; the third pleaded guilty to obstruction of justice in 2015[3].
…
The nature of Mallove's work led to some conspiracy theories[13] regarding the homicide, but police suspected robbery as the motive.[14][15]
Not for one second do I believe Mallove's death was a random "robbery"!
Note: Mallove also wrote that Fire From Water documentary, yet like always Wikipedia removes these details…
The documentary is an amazing work and it explains why the pathetic establishment don’t want to give it too much publicity and thus not include it in Mallove's Wiki page…
http://www.infinite-energy.com/whoarewe/gene.html
Retrieved: 15 September 2017
Archive: https://archive.is/507wE
Infinite Energy and New Energy Foundation founder Dr. Eugene Mallove was murdered on May 14, 2004. His death has left a tremendous void in not only the daily lives of those who loved him, but also within the new energy field. Gene was a long-time advocate for new energy science and technology and perhaps the most well-known spokesperson for the field.
We were unsure whether the magazine and non-profit could continue without Gene's energy and commitment. Ultimately, we decided that we had to make a go of it, to try to carry on with Gene's vision. The magazine continues to have a dedicated audience and even though Gene's voice in its pages is desperately missed, Infinite Energy continues to publish. With the support of our fantastic donors, the non-profit New Energy Foundation has also been able to dole out over $1 million in grants to new energy researchers or organizations since Gene founded the organization in 2003.
Wow… Gene gets murdered in 2004 a year after founding Infinite Energy and New Energy Foundation… # RIPGene
Given the fact that Dr. Judy Wood published her brilliant game-changing work "Where Did The Towers Go?" in 2010, Steven Jones' connection to Cold Fusion and 9/11, the likelihood of Judy Wood and Eugene Mallove joining forces to blow the entire lid off #FreeEnergy technology and 9/11 was too high a risk that the pathetic, yet extremely cunning, ruling party did not want to take…
http://wheredidthetowersgo.com/articles/cc/CB.html
Retrieved: 12 September 2017
Archive: https://archive.is/XVD7u
…
The following information clearly illustrates Steven Jones to be a government plant to discredit free energy in the public eye, and to mislead the 9/11 Truth Movement with deceitful research.
…
STEVEN JONES' RESEARCH LINKED TO TWO SUSPICIOUS MURDERS
In May 2004, Founder of Infinite Energy Magazine, Dr Eugene Mallove, wrote an open letter to the world entitled Universal Appeal for Support for New Energy Science and Technology”. In this letter, Mallove specifically mentions cold fusion as a possible alternative to oil and asks for research funds.
A few months later, Eugene was murdered by two people.
MES Note: The dates seem a bit off here as Eugene was murdered on 14 May 2004.
Three months before his death, Mallove was interviewed on the "Coast To Coast" radio program. This must-listen interview from February 3, 2004 can be downloaded in a 13MB MP3 here.
Dr. Mallove, a known supporter of P&F’s research, discussed a cold fusion patent that was approved a few weeks before 9/11.
Mallove discusses his book "Fire From Ice" and the emerging technology of cold fusion in this must-watch mid-1993 TV news interview clip.
More information on Eugene Mallove here.
Infinite Energy Magazine: Cold Fusion in the News
…
In March 2006, Dr Judy Wood and her student, Michael Zebuhr, performed demonstrations with molten aluminum at Clemson University to show that Steven Jones’ research was faulty.
Two and a half weeks later, Michael was murdered by two people.
After Michael was killed, Judy Wood received an email with the following message:
"we've done it before and we will do it again if need be."
***SIMILARITIES BETWEEN COLD FUSION AND 9/11 ***
*Jones used muon-catalyzed fusion to distract people
from P&F's electrochemically-induced process research.*Jones used thermate to distract people
from W&R's directed energy weapon research.
*Eugene Mallove was killed by two people
after writing an open letter asking for cold fusion research funds.
His letter interfered with Jones' deceptions so
his killing must have been a warning.*Michael Zebuhr was killed by two people
after doing research with molten aluminum.
His research interfered with Jones' deceptions so
his killing must have been a warning.
Cold Fusion = 9/11
Stanley Pons = Judy Wood
Martin Fleischmann = Morgan Reynolds
Electrochemically-induced Process = Directed Energy Weapons
Muon-catalyzed Fusion = Thermate
Eugene Mallove = Michael Zebuhr
http://drjudywood.com/articles/JJ/JJ7.html#mzebuhr
Retrieved: 12 September 2017
Archive: https://archive.is/Q7KQO
…
In Memory of Michael Zebuhr
September 21, 1980 - March 19, 2006

Figure 19. This picture was taken by Michael Zebuhr (Clemson University) on 28 Feb 2006, to demonstrate that aluminum does indeed glow, if hot. Here, the aluminum is on a tungsten "boat" between two electrodes. The boad is heated by an electrical current. At this temperature, an increase in the temperature would increase the brightness and white-out the picture similar to photographing an incandescant light.
Figure 20: The team casts a crankcase from aluminum.
PHOTO BY JUDY HAY
source: Popular MechanicsThe molten aluminum is glowing.
The environment is at atmospheric pressure.
The lighting is "daylight conditions" (outside).Here is the last 20 minutes of Jim Fetzer's radio show (10 Aug 2006)
with Steven Jones, who claims aluminum doesn't glow. (The full show is hour1 and hour2.)Why do we have an educated nuclear physicist emphatically denying that aluminum glows? The mystery has now been solved! (See ___)
As with all metals, emissivity varies with temperature, as explained here.(*) In Memory of Michael Zebuhr
September 21, 1980 - March 19, 2006(3/1/06) Aluminum Glows
(*) Justice for Michael Zebuhr
Following the murder of my student, Michael Zebuhr, a truly extraordinary human being, I received an email telling me, "we've done it before and we will do it again if need be." Michael told me, "Whatever happens, don't ever stop pursuing this. It's too important."
Had Michael not been murdered, how long would it have taken for him to make the connection between his uncle (Bill Zebuhr), Gene Mallove, and Steven E. Jones?
The stakes are high but we must all never stop pursuing this!
http://www.infinite-energy.com/iemagazine/issue56/memorial.html
Retrieved: 12 September 2017
Archive: https://archive.is/N4IGa
We have all suffered a great loss. Gene Mallove was killed on the evening of Friday, May 14. He was at his childhood home in Norwich, Connecticut, cleaning up between tenants, when he was murdered in an apparent robbery. Infinite Energy will provide reports as more details become available.
…
Gene asked me to be Chairman of the New Energy Foundation (NEF) Board of Directors because of my background and my strong interest in Infinite Energy Magazine and the peripheral projects he was involved in.
Looks like Mallove and Zebuhr were hitting a bit too close to the truth…
Summary and Conclusions
This is an extremely long introduction to Nuclear Physics, but hopefully this will help connect some much needed dots into developing #FreeEnergy technology!
Here is a summary of my understanding of Mainstream Nuclear Physics:
Atoms, Ordinary Matter, Atomic Nucleus, Protons, Neutrons, Electrons, Isotopes
- Atoms are the smallest unit of ordinary matter.
- Ordinary matter comprises 15% of the Universe; the unknown remainder is Dark Matter.
- Dark Energy is the term used for the source of the accelerating expansion of the universe.
- Dark Energy makes up 73% of the energy of the universe.
- Dark Energy and Dark Matter make up 96% of the energy of the universe.
- Effectively, modern physics knows little to nothing of 96% of the universe.
- Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.
- Plasma is a gaseous mixture of negatively charged electrons and highly charged positive ions which are distinctly separated such that electric fields, currents, and magnetic fields are created; such as lightening.
- Atoms are subject to quantum effects.
- Atoms have a nucleus.
- Atoms have 1+ electrons bound to the nucleus.
- Nucleus is made of 1+ protons and similar number of neutrons.
- Protons and neutrons are called nucleons.
- 99.94% of an atom's mass is in the nucleus.
- Protons have a positive electric charge.
- Electrons have a negative electric charge.
- Neutrons have no electric charge.
- If number of protons and electrons are equal, that atom is electrically neutral.
- Non-neutral atoms have an overall negative or positive charge, and are called ions.
- Electrons attracted to nucleus by the electromagnetic force.
- Protons and neutrons are attracted to each other by the nuclear force.
- Nuclear decay / Nuclear transmutation occurs when the repelling electromagnetic force exceeds the nuclear force, resulting in nucleons ejecting from the nucleus.
- Number of protons defines chemical element of the atom.
- Number of neutrons defines the isotope of the element.
- Number of electrons defines magnetic properties of atoms.
- Chemical bonds between atoms forms molecules.
- Branch of physics studying the Atomic Nucleus is known as Nuclear Physics.
Quantization, Quantum Mechanics, Particle-Wave Duality
- Energy, Momentum, and other quantities are restricted to certain discrete values, such as the charge of an electron, and this referred to as quantization.
- Quantum objects behave both as particles and waves.
- The act of observing affects the observations themselves.
- Physical properties of quantum objects are based on probability rather than finite measurements.
Fundamental Forces: Gravity, Electromagnetism, Strong, Weak
- There are 4 known fundamental forces or interactions that appear to be irreducible: Gravitational, Electromagnetic, Strong, and Weak Forces.
- Strong and Weak Forces or Interactions are also known as Strong Nuclear and Weak Nuclear Forces or Interactions.
- It is theorized that all forces are related in one "Theory of Everything".
- Gravity is a force by which all objects with mass (and hence energy) move towards each other.
- Gravity is best described by curvature of spacetime due to uneven mass distribution.
- Black holes are extreme cases of this curvature in which nothing can escape its horizon, including light.
- The Electromagnetic Force is carried by the photon and occurs between electrically charged particles.
- Electricity and magnetism are different manifestations of the same interaction.
- The Strong Force is carried by gluons to bind quarks together to form hadrons such as protons and neutrons.
- The Strong Force has a residual Nuclear Force that binds the hadrons to form atomic nuclei.
- The Weak Force is carried by W and Z bosons and mediates radioactive decay.
- Particle accelerators use electromagnetic fields to propel charged particles at near light speed in a vacuum.
- They are often used as colliders and other applications to better understand the structure of the subatomic world.
Subatomic Particles, Elementary Particles, Fermions, Bosons, Gluons
- Subatomic particles are particles much smaller than atoms.
- Two Types of Subatomic Particles: Elementary Particles and Composite Particles.
- Elementary particles have an unknown substructure.
- Two Types of Elementary Particles: Fermions and Bosons
- Fermions consist of elementary particles of matter.
- Bosons consist of elementary particles of force which mediate interactions between fermions.
- Spin is an intrinsic form of angular momentum carried by atomic nuclei and subatomic particles.
- The Higgs Boson is a special type of boson that has no spin and doesn't carry force; known as a "Scalar boson".
- Electrons are fermions.
- Protons are subatomic hadron particles.
- Neutrons are subatomic hadron particles.
- A Hadron is a composite particle made up of quarks held together by the strong force.
- A quark is a fermion.
- Quarks are only known through observations of hadrons.
- There are six types of quarks, known as flavors.
- Gluons are bosons that mediate the strong force between quarks.
- Photons are bosons that mediate electromagnetic interaction.
- W and Z Bosons mediate Weak Interaction.
- Graviton is a hypothetical boson that mediates Gravity.
Antiparticles, Antimatter
- Most particles have an associated antiparticle with the same mass (in theory) but opposite charge.
- These antiparticles are very difficult to produce and difficult to contain.
- Their existence is shown by particle accelerators/colliders.
- Antimatter is a material made up antiparticles.
- For the most part Antimatter remain a mystery, at least to me.
- Particles can collide with antiparticles releasing large amounts of energy.
- Visible universe is mostly ordinary matter, which make this asymmetry of matter make up one of the biggest unsolved problems in physics. (The biggest is obviously 9/11).
- Electrons and Positrons (antielectrons) can be denoted as e- and e+, respectively.
Electromagnetic Field, Electric Field, Magnetic Field
- An electromagnetic field (EMF or EM field) is a physical field extending throughout space which is produced by electrically charged objects.
- An electric charge is a physical property of matter that causes it to experience a force when inside an EMF.
- Like charges attract, opposite charges repel.
- EMF is a combination of electric and magnetic fields.
- Can be viewed as a continuous structure or as quanta of fixed frequencies/wavelengths, in this case of photons.
- An electric field is a vector field (has magnitude and direction) of Coulomb forces (force between static electrically charged particles).
- Electric fields are created by electric charges or time-varying magnetic fields.
- A magnetic field is a vector field of force that pull "magnetic" objects.
- Magnetic fields are produced by moving electric charges (current), hence Right Hand Rule: Thumb = current; Fingers = B-Field.
- Since elementary particles may also have electric charges, spin, and move around they also create their own magnetic field.
- Magnetic fields move from "North" to "South", hence a compass pointing north is actually "seeking south"; thus Earth's North Pole is actually a south pole! # MindBlown
- Magnetic B field is generated by currents whereas the H field is another field to account for the internal magnetic fields of materials within the B field.
**Electromagnetic Radiation, Photon Energy, Light **
- EM radiation or EMR are the waves or, in quantum mechanics, their quanta (photons), of the EMF and carry radiant energy (also referred to as photon energy).
- The term "radiation" is to exclude static electric and magnetic and near fields.
- Near fields are regions of an EM Field close to an object or charged particle that produced it, thus being influenced by that object.
- EMR can be viewed as the Far Field as it "radiates" away from the object/charge that produced it.
- EM waves are composed of oscillating perpendicular electric and magnetic fields.
- Photon energy is the function of its EM wavelength and move in the direction of the EM wave.
- The longer the wavelength, the lower its energy.
- The higher the frequency, the higher the energy.
- Photons are considered massless yet have energy (same as gluons), which raises some issues regarding Einstein's Mass-Energy equivalence formula E = mc2!
- EMR consists of EM waves which are synchronized electric and magnetic fields that propagate at the speed of light in a vacuum.
- EM waves are produced whenever charged particles are accelerated (i.e. increase in velocity) and can interact with other charged particles.
- EM waves in order of increasing frequency/decreasing wavelength/increasing energy: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, and gamma-rays.
- Speed of light is defined as constant and the meter is defined based on the speed of light.
- Light sometimes refers to all EMR.
- Visible light makes up a tiny fraction of the total light.
- Different frequencies of visible light make up the colors of a rainbow.
- White light occurs when all the visible light frequencies are equally present ("white" can be referred to other wave-types, i.e. "white sound").
- A dispersive prism separates the different wavelengths of light so that that they are scattered and thus their colors are visible.
Gamma Radiation, Radioactive Decay, Ionizing Radiation
- Gamma rays/radiation are EMR in the highest observed energy range.
- They arise from radioactive decay of atomic nuclei, in this case called Gamma decay, and usually after other types of decay have occurred.
- Gamma rays can ionize atoms.
- Radioactive decay (or nuclear decay) is the process in which an unstable atomic nucleus loses energy by emitting particles out.
- Large elements are usually radioactive.
- Ionizing radiation is when radiation has enough energy to liberate electrons from atoms or molecules, thus making them ions.
- EMR of the higher part of UV rays, X-rays, and Gamma rays are ionizing.
- Lightning strikes are rare natural sources of gamma rays not of a nuclear origin. # VeryInteresting.
- Gamma rays are emitted during nuclear fission.
- Gamma rays can only be reduced by thick mass such as a very thick layer of lead.
- Alpha decay is when a helium nucleus is emitted from an atomic nucleus.
- Beta decay is when an electron (ß+ decay) or positron (ß- decay) and a neutrino (a type of fermion) is emitted from an atomic nucleus.
Nuclear Chain Reactions, Nuclides, Deuterium, Tritium
- A nuclear reaction is when two atomic nuclei or one nuclei and an outside subatomic particle collide to produce one or more nuclides that are different from the starting nuclide(s).
- A nuclear chain reaction is when a nuclear reaction results in more nuclear reactions giving rise to a possibility of a self-propagating series of more nuclear reactions.
- If the collision doesn't involve changing the nature of the nuclide, then this is referred to as nuclear scattering.
- Nuclides refer to nuclei characterized by its number of protons, neutrons, and nuclear energy state.
- Deuterium or 2H is a stable isotope of Hydrogen, and has 1 proton and 1 neutron.
- Tritium or 3H is an unstable and much more naturally rare isotope of Hydrogen, and has 1 proton and 2 neutrons.
- Note: the 2 and 3 above refer to the Atomic Mass Number: Number of Protons + Number of Neutrons.
- Deuterium and Tritium are used in nuclear fusion applications.
Nuclear Fission, Fusion, and Transmutation; Quantum Tunneling
- Nuclear Fission is the process of a nucleus splitting into smaller lighter nuclei.
- Fission occurs either by a nuclear reaction or a natural radioactive decay process.
- Fission process produces free neutrons, gamma photons, and releases a very large amount of energy.
- Nuclear Fusion is when nuclei come close enough to each other to form one or more different nuclei.
- The difference in masses of reactants and products is represented by a large amount of energy released.
- Fusion that produces "light" elements usually has a net energy release.
- Fusion that produces "heavy" elements usually results in a net energy retention.
- Opposite is true for Fission.
- Nuclear Transmutation is the conversion of one chemical element or its isotope into another; hence when the number of protons or neutrons changes.
- Nuclear Fission and Fusion are both types of Transmutation.
- Fission process results in dangerous radioactive nuclear waste.
- Fusion process results in much less radioactive nuclear waste.
- Nuclei need to be brought close enough together in order for Quantum Tunneling to occur and overcome repulsive electrostatic forces.
- Quantum Tunneling is the term used to describe the phenomenon of particles seemingly moving past a barrier that classical physics can't explain.
Nuclear Weapons, Fission Bombs, Thermonuclear Bombs, Contamination
- Nuclear Weapons are formed from Nuclear Reactions: Fission or a combination of Fission and Fusion termed Thermonuclear.
- Large amount of energy is released from small amounts of matter.
- Fission bombs (or A-Bombs) involve forcing fissile material into supercriticality.
- Supercriticality is achieved by shooting subcritical masses (via explosives) at each other (Gun-type) or by compression with explosives (Implosion-type).
- A subcritical mass is a fissile material that can't sustain a fission chain reaction.
- A critical mass is a fissile material self-sustaining a fission chain reaction.
- A supercritical mass is when the material has an increasing rate of nuclear chain reactions.
- Thermonuclear bombs use fission reactions to trigger fusion reactions.
- Most nuclear weapons use the thermonuclear design because it is more efficient.
- A Hydrogen Bomb is a type of Thermonuclear bomb that involves fusing hydrogen isotypes like deuterium and tritium.
- Fusion reactions create far less fission products.
- The radioactive contamination from both types of bombs are very large because both have at least one fission stage.
Manhattan Project, World War 2, Hiroshima, Nagasaki, Office of Censorship
- The Manhattan Project was a top secret project during World War 2 by the United States with support from the UK and Canada to develop the first nuclear weapons.
- The Manhattan Project employed over 100,000 thousand employees in 3 completely manufactured hidden cities (Oak Ridge, Hanford, and Los Alamos) in which nearly everyone involved was clueless as to what their work was actually leaning towards.
- The Los Alamos Laboratory in New Mexico, USA was tasked with designing and building the atomic bombs.
- The Manhattan Project, along with 9/11, is undeniable proof that large-scale conspiracies can and will happen.
- The Office of Censorship was established during the war to censor all communications coming inside and outside of the United States.
- Even before the Office of Censorship and before the war, the American press "voluntarily" censored itself after the Pearl Harbor Bombing. # DemocracyDiesInDarkness
- The Manhattan Project, along with advancements in Radar technology, were the major topics being censored from the public.
- Hiroshima, Japan was bombed by the first ever Nuclear Bomb, a uranium gun-type fission bomb named "Little Boy" on August 6, 1945.
- Nagasaki, Japan was bombed by a plutonium implosion-type fission bomb named "Fat Man" on August 9, 1945. # NeverForget
- The Manhattan Project, and the United Nations, was likely first hatched at the Bohemian Grove in San Francisco where rich powerful men meet annually in secret to perform a ritual mock human sacrifice to a giant owl statue… # ThisIsWhatTheElitesThinkOfUs # WakeUp # NoMoreWar
Nuclear Reactors, Fission Reactors, Fusion Reactors, Fukushima
- Nuclear reactors initiate and control a nuclear chain reaction to obtain useful energy.
- All commercial nuclear power reactors are based on nuclear fission.
- Two classes of fission reactors: Thermal and Fast neutron reactors.
- Thermal reactors are the most common and use a moderator material (such as water) to slow down neutrons to control the fission of their fuel.
- Fast neutron reactors use fast or higher energy neutrons to cause fission.
- Nuclear Fusion reactors is an experimental technology due to the high energy/heat required for Fusion.
- Fukushima, Japan nuclear disaster occurred on 11 March 2011 in which a tsunami following an earthquake disabled control of the Daiichi nuclear power plant.
- Questions as to whether the accident was foreseeable, and whether even advanced countries like Japan can handle nuclear power were raised; and the ideas of conspiracy rise as well as they should in a post 9/11 world. # NeverForget
Cold Fusion, Pons/Fleischmann, Patents, Perpetual Motion
- Cold Fusion is nuclear reactions or fusion at or near room temperature.
- The conventional nuclear reactions thus far discussed are considered "hot" fusions.
- Current "accepted" theory suggests cold fusion is impossible.
- World-renowned electrochemists Martin Fleischmann and Stanley Pons claim to have produced nuclear reactions by a small tabletop experiment involving an electro-chemical reaction with heavy water and palladium.
- They hypothesized that palladium metal can absorb deuterium, thus resulting in nuclear fusion to form helium.
- They reported trace byproducts such as neutrons and tritium.
- They reported little to no dangerous radiation; i.e. gamma rays.
- Their findings raised hopes of clean #FreeEnergy!
- Ultimately they were ridiculed by the "scientific" community, the mainstream media, and the United States government avoided funding for cold fusion.
- In fact, the University of Utah asked Congress for $25 million to continue further research into Cold Fusion, and Pons & Fleischmann scheduled a meeting with representatives of President George Bush Sr.
- Fake news outlets like the New York Times & the Boston Herald and fake science institutions such as Caltech Institute of Technology & CERN ridiculed Pons and Fleischmann so much that the meeting with Washington Representatives were cancelled… # NWO
- Government disinfo-tool Steven Jones is instrumental in covering up Cold Fusion and creating the controlled opposition group Architects & Engineers for 9/11 Truth in order to cover up 9/11 #FreeEnergy tech # NotACoincidence
- United States doesn't accept patents claiming Cold Fusion or Perpetual Motion Machines.
- Pons and Fleischmann NEVER retracted their claims.
- Mainstream "science" institutions ridicules and defunds anyone willing to research into Cold Fusion.
- Many independent researchers, including yours truly, are doing the work that the coward "scientists" should be doing! # SomebodyGottaDoIt
- Pons and Fleischmann's only mistake was they had no idea about the technological global dictatorship that we all live in, until it was too late…
Eugene Mallove and Michael Zebuhr's Murders, Dr. Judy Wood Threatening Email
- Eugene Mallove had science degrees from both MIT and Harvard University.
- Mallove was the author of the 1999 book Fire From Ice: Searching for the Truth Behind the Cold Fusion Furor
- Mallove co-wrote the 1998 documentary Cold Fusion: Fire From Water.
- Mallove founded the organizations Infinite Energy and New Energy Foundation in 2003.
- Mallove was a strong and well-known proponent of new energy science and helped exposed the Cold Fusion coverup, which the infamous Steven Jones was a part of.
- Mallove was murdered in 2004.
- Michael Zebuhr was Dr. Judy Wood's student.
- Zebuhr was performing experiments on molten aluminum showing it glowed to disprove 9/11 controlled opposition Steven Jones' assertion that glowing flowing metal during 9/11 could not have been aluminum.
- Zebuhr was murdered in 2006.
- Dr. Judy Wood then received a threatening email stating "we've done it before and we will do it again if need be"…
- Zebuhr's uncle was Bill Zebuhr, CHAIRMAN of the New Energy Foundation founded by Eugene Mallove… # YouCantMakeThisStuffUp!
- Dr. Judy Wood publishes here monumental work "Where Did The Towers Go?" in 2010.
- It is my opinion that Mallove and Zebuhr ALMOST CERTAINLY were killed to ensure that the #FreeEnergy movement is fractured because the combination of Zebuhr, Mallove, and Judy Wood would have the shook the foundations of the current evil ruling establishment.
This super-long video is a way for me to obtain an overview of mainstream nuclear physics, and to show just what happens to people that stray away from the mainstream…
This is why we must all wake up together, educate ourselves, self-govern ourselves, and ensure that #FreeEnergy technology is not only made available but that safeguards are discovered so that technology never becomes hidden in the first place!
Unlike many online #FreeEnergy researchers, I don't make any claims of grandeur, and I am willing to put in the effort to truly uncover the science and technology hidden from us, so please donate through Patreon, PayPal, or Bitcoin at https://mes.fm/donate so that I can work full time on this project!
Stay Tuned For #FreeEnergy Part 3…