Hello friends of Steemit! Continuing with the series of posts on diffusion in porous media, this time I will talk about some types of diffusion in porous media. Maybe you are interested in reading the previous posts on the subject, so at the end of the article I will leave the links to them. In such posts, I talk about the diffusion and the equations that describe it and about the porous media.
Figure 1: Diffusion of a particle in a two-dimensional porous medium
In porous media, heterogeneous materials formed by a solid phase with empty spaces called pores (like a sponge, the soil, the skin), the diffusion is a complex phenomenon, and "can be carried out in three different ways: ordinary diffusion, Knudsen diffusion and surface diffusion" [1].
ORDINARY DIFFUSION
The ordinary diffusion occurs if the pores are very large compared to the mean free path of the particles of the diffused substance, that is, when the average pore size is much larger than the mean free path of the particles, this being the average distance traveled by the particles before colliding. Mathematically, such condition is expressed as:
where:


In the case of ordinary diffusion in porous media, the diffusive flow is described as a function of the effective diffusion coefficient or effective diffusivity, by means of the first Fick's equation.
where:

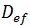

is the gradient of the concentration of the substance, or change in concentration per unit length.
In the case of models of porous media models with capillaries, it has been found [1, 2] that the effective coefficient of diffusion or effective diffusivity depends on the porosity and the tortuosity (defined as the ratio or the quotient between the length of the path followed by the particles and the minimum length between the two points of the medium) in the following way:
where:



KNUDSEN DIFFUSION
The Knudsen diffusion occurs if the mean free path of the diffused particles is greater than the average pore size, that is, when:
In this type of diffusion, the transfer rate of the particles is highly influenced by the collisions with the walls of the pores. The Knudsen diffusion appears when:
The Knudsen diffusion "is predominant in pores whose average diameter varies between 2 nm and 50 nm" [4, 5] (nm is the abbreviation of nanometer, and is equivalent to a billionth of a meter; small!, right?), that is, it is the predominant type of diffusion in nanoporous and nanostructured materials, of which I will speak in future posts, since they are currently of great interest for the advance of nanotechnologies.
The danish physicist Martin Hans Christian Knudsen (1871 – 1949) "showed that, under these conditions, the diffusive flow of a gas in a capillary" [3], is given by:
with:
where:




is the fraction of molecules colliding with the walls of the capillary, which is generally taken equal to one.
It has been found experimentally [1], that effective Knudsen diffusivity is given by:
A greater dependence of the Knudsen diffusivity is observed in relation to the porosity than in the case of the effective diffusivity for the case of molecular diffusion.
SURFACE DIFFUSION
The surface diffusion occurs when molecules that have been absorbed by a porous medium are transported across the surface of the porous as a result of a two-dimensional or surface gradient of concentration.
Generally, the surface diffusion plays a minor role in the diffusion in a porous medium unless there is a large amount of absorption by the solid phase of the medium.
Although, the three types of diffusion mentioned, are the most common in porous media, there are other types of diffusive processes in porous media included under the term anomalous diffusion.
I hope you enjoyed the publication about the research in which I work and that the information is your useful, I will gladly clarify your doubts and concerns about the subject. Thanks for your kind reading.
I wait for you in the next installment, where I will talk about the procedure I followed in my researchs to simulate the phenomenon of diffusion in porous media.
If you are interested, I leave the link of the previous article of the series:
Diffusion in Porous Media – A world of applications – Part 1
Diffusion in Porous Media – A world of applications – Part 2
Diffusion in Porous Media – A world of applications – Part 3
Note: The Figure 1 is self-made.
References:
[1] Welty, J. R.; Wicks, C. E. y Wilson, R. E., 1984. Fundamentos de transferencia de momento, calor y masa, Edit. Limusa, México D.C., México, pp. 533-733.
[2] Krasuk, J. H., 2002. Transferencia de masa por difusión, Ediciones MCA, Los Teques, Venezuela, pp. 1-175.
[3] Youngquist, G. R., 1970. Diffusion and flow of gases in porous media. EN: Flow through porous media, American Chemical Society Publications, Washington D.C., EUA, pp. 57-70.
[4] Malek, K. and Coppens, M. O., 2003. Knudsen self- and Fickian diffusion in rough nanoporous media. Journal of Chemical Physics. 119(5): p. 2801-2811.










